Cover photo by Nick Evans.
Find the Long-toed Tree Frog in the FBIS database (Freshwater Biodiversity Information System) here.
Family Heleophrynidae
Identification
The Long-toed Tree Frog is a large frog, reaching 50 mm in snout–vent length. It has a uniformly green dorsum and a creamy white belly. It lacks the dark mask of L. mossambicus, and the dorsal mottling that is common in L. natalensis. Other characters that distinguish L. xenodactylus from the other two species in the atlas region are its longer toes and fingers with less distinct discs at their tips. The size of the inner metatarsal tubercle and extent of webbing between the toes are intermediate between those of the other two species (Poynton 1963; Van Dijk 1978b; Lambiris 1989a; Passmore and Carruthers 1995). The advertisement call consists of one or two short, pulsed croaks uttered at long intervals. Each croak is 0.1 s in duration, has a dominant frequency of about 1000 Hz, and is sometimes preceded by a soft buzzing (Passmore and Carruthers 1995).

Ntsikeni Nature Reserve, KwaZulu-Natal
Photo by Nick Evans
Habitat
This species occurs in the Grassland Biome, mainly in high altitude Moist Upland Grassland that receives annual summer rainfall of 650–1000 mm. It also inhabits Short Mistbelt Grassland and North-eastern Mountain Grassland (Lambiris 1989a; Harrison et al. 2001). It is predicted to occur in high-rainfall central and western grasslands of KwaZulu-Natal (Armstrong 2001). Most populations occur at altitudes of c.1000 m, but the species is also recorded at 1830 m in the uKhahlamba-Drakensberg Park (Giant’s Castle; Lambiris 1989a).
The Long-toed Reed Frog breeds in grassy wetlands and marshes. At Mpur forestry area (3029BC), the wetland consists of a maze of eroded channels with many small, grass-covered islands.
Behaviour
Calling has been recorded in December and January. At the Mpur forestry area, males call from well-concealed positions in grass tussocks and from burrows (Passmore and Carruthers 1995), but also from the tops of grass-covered islands (pers. obs.). A captive specimen remained buried in the soil during the day, with only its head visible, and emerged and became active at night (Van Dijk 1978b). In the wild, these frogs are known to climb vegetation (Channing 2001), but field observations suggest that they are the least arboreal of the three southern African Leptopelis species.
The biology of this species is still poorly known, and its eggs and tadpoles have not been described.

Merthley Wetland, KwaZulu-Natal
Photo by D Rollinson
Status and Conservation
Status
Previously listed as Rare (McLachlan 1978), Restricted (Lambiris 1988) and Vulnerable (IUCN 2000), L. xenodactylus is now classified as Endangered (Harrison et al. 2001; this publication). This status was assigned because of the species’ small area of occupancy, severely fragmented distribution, and evidence of continuing decline in the area of occupancy, extent and quality of habitat, and number of locations (Harrison et al. 2001).
The Long-toed Tree Frog occurs in the uKhahlamba-Drakensberg Park (Giant’s Castle and Highmoor sections), Weza Forest Reserve, and the Minerva Private Nature Reserve near Richmond. It is regionally listed as a “species of conservation importance” by the KwaZulu-Natal Nature Conservation Service, and is protected in that province in terms of the KwaZulu-Natal Nature Conservation Ordinance, Number 15 of 1974, as amended.
Threats
The main threat to L. xenodactylus populations is the degradation, fragmentation and loss of habitat caused by afforestation and infestations of alien plants. Of concern too is the lack of information regarding the general biology of the species, that makes it difficult to assess limiting factors and conservation needs.
Recommended conservation actions
Surveys are needed to establish the complete distribution of L. xenodactylus. To this end, Armstrong’s (2001) predictions of its potential distribution provide a useful starting point. Furthermore, the results of surveys would help to determine the predictive power of the model used. Research effort should also be focussed on life history and the identification of limiting factors. A Population and Habitat Viability Assessment (PHVA) is recommended. Known populations should be monitored and protected from further habitat loss or degradation.
It is recommended that the locality at Mpur forestry area be recognized as a site of special importance (Burger and Harrison 2002), and that it be protected and managed accordingly by Singisi Forest Products and Eastern Cape Nature Conservation Department.
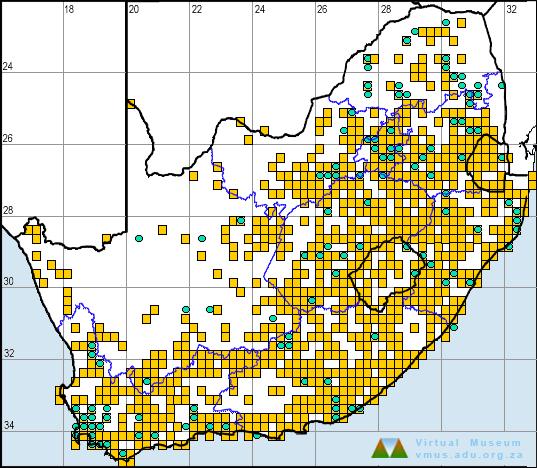
Distribution
The Long-toed Tree Frog is endemic to the atlas region, occurring mainly in the southern KwaZulu-Natal highlands, but also marginally in adjacent parts of the Eastern Cape Province. The species was described from a single specimen collected 40 years ago at Underberg (2929CD), and 15 years elapsed before a second specimen was found at Mpur forestry area near Franklin (3029BC; Van Dijk 1978b). Currently, it is known from only nine quarter-degree grid cells, spanning the area from Weza (3029DA, DB) in the south to Giant’s Castle (uKhahlamba-Drakensberg Park; 2929AB) in the north. However, Armstrong (2001) has predicted a substantially larger range for this species. Note that the Franklin (3029AD) record as plotted by Lambiris (1989a) is incorrect. The correct location of this record is Mpur forestry area (3029BC). No records are currently known from the Franklin grid cell.
During the atlas period, L. xenodactylus was recorded at only five localities, three of which represent new distribution records. The Mpur forestry area (3029BC) is of special importance because it currently supports the largest known population of L. xenodactylus (at least 50 males), as well as the Critically Endangered Arthroleptella ngongoniensis (Burger and Harrison 2002; pers. obs.). The atlas data are accurate but incomplete.

Further Resources
The use of photographs by Dominic Rollinson and Nick Evans is acknowledged.
Long-toed Tree Frog Leptopelis xenodactylus Poynton, 1963
Other Common Names: Langtoonboompadda (Afrikaans)
Recommended citation format: Burger, M; Tippett, RM. (2025). Long-toed Tree Frog Leptopelis xenodactylus. Biodiversity and Development Institute, Cape Town. Available online at https://thebdi.org/2021/11/15/long-toed-tree-frog-leptopelis-xenodactylus/
This species text has been updated and expanded from the text in the
2004 frog atlas: Burger, M. (2004). Long-toed Tree Frog Leptopelis xenodactylus. In Minter LR et al 2004.
References:
Minter, LR; Burger, M; Harrison, JA; Braack, HH; Bishop, PJ; Kloepfer, D. (Editors). (2004). Atlas and Red Data Book of the Frogs of South Africa, Lesotho and Swaziland. Smithsonian Institution, Washington, and Avian Demography
Unit, Cape Town.
Carruthers, V; du Preez, L. (2017). Frogs of southern Africa: A Complete Guide. Struik Nature, Cape Town.
Channing, A. (2001) Amphibians of Central and Southern Africa. Protea Book House, Pretoria
Claus, B; Claus, R. (2002). Common Amphibians and Reptiles of Botswana. Gamsberg Macmillan, Windhoek.