Cover photo by Luke Kemp.
Find the Striped Caco in the FBIS database (Freshwater Biodiversity Information System) here.
Family Pyxicephalidae
Identification
The Striped Caco is a small species that does not exceed 21 mm in snout–vent length. It is readily distinguishable from its congeners by differences in colouration and vocalizations. The dorsum is light brown to orange-brown or even green, with a pair of dark brown dorsolateral stripes that extend from behind the eye to the groin. The flanks and dorsal surfaces of the limbs are somewhat spotted, while the ventrum is generally immaculate white, although sometimes marked with pale grey blotches. C. striatum may be distinguished from C. boettgeri and C. nanum by the presence of a light line that extends from heel to heel along the posterior surfaces of thighs, as well as a series of small, slightly elongated paravertebral glandular ridges, and a longer pair associated with the dorsolateral stripes over the anterior two-thirds of the body (FitzSimons 1947). The carpal tubercles are less prominent than those of C. boettgeri (Lambiris 1989a).

Eastern Shores, iSimangaliso Wetland Park, KwaZulu-Natal
Photo by Ryan Tippett
The advertisement calls of nine individuals recorded at Sneezewood Flats, Mpur forestry area (3029BC), consist of two components: short chirps and protracted “creaks” (M. Burger pers. comm.). Call bouts usually begin with several “chirps” (e.g., single, then double, then triple) and culminate in a series of creaks repeated up to a dozen times, but probably more in strong choruses. The chirps have a duration of 26–111 ms (mean 61) and an emphasized frequency of 5–5.5 kHz (mean 5.3), and consist of 4–14 pulses, emitted at a pulse rate of 101–187 pulses/s (mean 141). The creaks have a duration of 245–535 ms (mean 362) and an emphasized frequency of 4.6–5.7 kHz (mean 5.3), and consist of 14–24 pulses, emitted at a pulse rate of 27–82 pulses/s (mean 53).
The distribution of museum specimens of C. striatum suggests the presence of separate high- and low-altitude forms. The presence of other frog species, such as Leptopelis xenodactylus, Amietia vertebralis, and Strongylopus hymenopus, that are endemic to the same restricted range as the high-altitude form of C. striatum, suggests that some selective forces may have operated (or vicariance events occurred) in this region, resulting in speciation.
The Striped Caco is one of the least studied species in the South African frog fauna. Poynton (1964) noted that the original description of this species did not adequately separate it from the highly variable C. boettgeri and placed it in the synonymy of the latter species. Lambiris (1989a) resurrected C. striatum on the basis of differences in laryngeal and buccopharyngeal morphology and identified diagnostic external morphological characters that separate the species from C. boettgeri. He also noted that the call of striatum differed from that of boettgeri, but was unable to provide a detailed description of the call. A subsequent revision of the genus Cacosternum, using morphological, osteological and genetic data, has confirmed the validity of C. striatum (E. Scott in prep.).
Habitat
The Striped Caco inhabits various vegetation types within the Grassland Biome in the summer-rainfall region. Breeding habitat appears to be in inundated wetlands, or adjacent to slow-flowing sidewaters of highland streams (FitzSimons 1947; Lambiris 1989a). The habitat of C. striatum conforms to that of C. boettgeri, making any ecological distinction difficult to identify. C. striatum has not been collected at the same site as C. boettgeri, although both C. boettgeri and C. striatum have been collected syntopically with C. nanum.
Behaviour
Almost no data is available on the life history of the Striped Caco. Male vocalization has been recorded during December, January and early February (M. Burger, H. Braack and A. Turner pers. comm.). Calling starts during the late afternoon and continues through the night. As is the case in C. boettgeri, the males call from well-concealed sites in dense grass tussocks at or just above the water-level.
Status and Conservation
Status
The Striped Caco was not listed in previous SA National Red Data Books, but was assigned to the category Data Deficient by Harrison et al. (2001), based on a lack of knowledge about the species’ taxonomic status, general biology and distribution.

Eastern Shores, iSimangaliso Wetland Park, KwaZulu-Natal
Photo by Ryan Tippett
C. striatum has been recorded from a number of protected areas, including Sehlabathebe National Park in Lesotho, SA Natural Heritage Site No.208 at Gilboa Estates (SAPPI) near Mooi River (2930AB), Cobham and Vernon Crookes nature reserves, and the iSimangaliso Wetland Park. Efforts are currently underway to protect the populations at Sneezewood Flats in the Mpur forest area (3029BC), under the management of Singisi Forestry Products. All amphibians are protected by KwaZulu-Natal Nature Conservation Ordinance, No. 15 of 1974, as amended.
Threats
In the grassland habitat occupied by the Striped Caco, afforestation and agriculture (e.g. sugar cane) pose the major threats to fauna and flora, and have resulted in the loss, decrease in quality, and fragmentation of its habitat.
Recommended conservation actions
At the time of writing, high priority is being given to clarification of the taxonomy, distribution and area of occupancy of C. striatum, as well as a detailed study of its life history and ecology. This should allow for the formulation of appropriate recommendations to the relevant conservation authorities in the near future.
Distribution
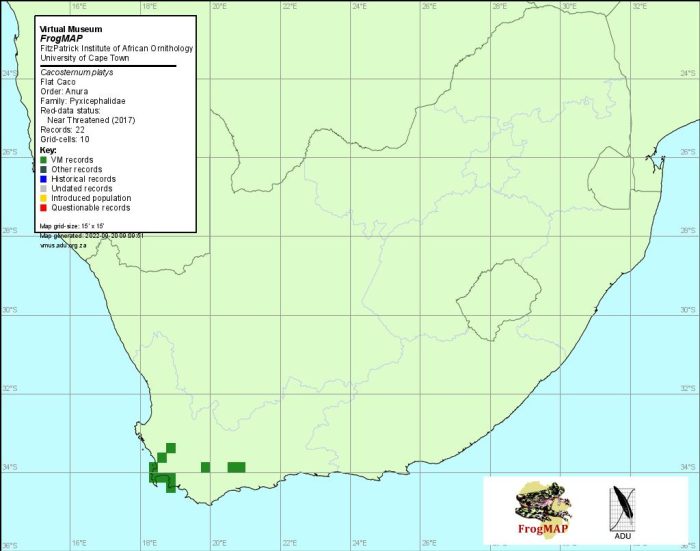
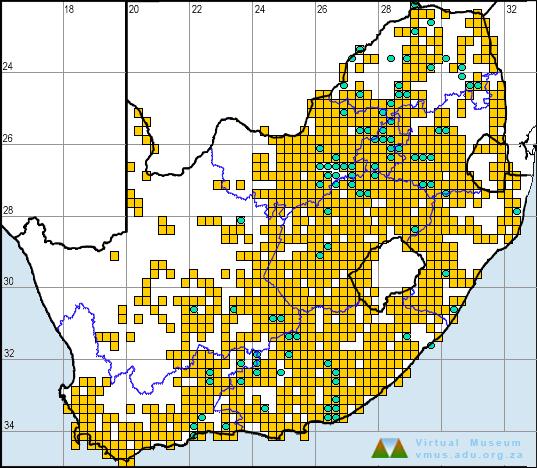
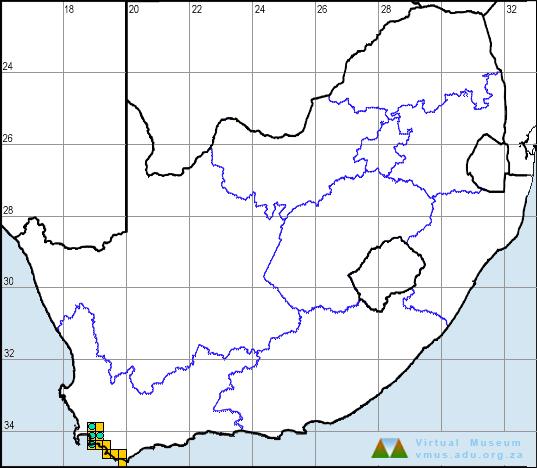
Low altitude populations of the Striped Caco (50–100 m) include the type locality, a golf course in Durban (2930DD), Charter’s Creek near St Lucia (2832AB), and KwaMbonambi and vicinitiy (2832CA) – all coastal areas with warm tropical climates. Two gravid females that fit the description of C. striatum better than they do any other species of Cacosternum, were collected at a slightly higher elevation (300–450 m) in Vernon Crookes Nature Reserve (3030BC).
The remaining material was collected at much higher altitudes (1500–2200 m) in the KwaZulu-Natal midlands at Cedarville (3029AC), Mpur forestry area east of Franklin (3029BC), Gilboa (2930AB), Impendhle Hills and Boston (2929DB), and in the Drakensberg at Kamberg Nature Reserve (2929BC) and Sehlabathebe National Park, Lesotho (2929CC). A single specimen was collected at an altitude of 1600 m at Matatiele (3028BD).
The data on the map are subject to change pending two studies in progress at the time of writing that are expected to clarify the taxonomic status and distribution of this species (E. Scott, M. Burger unpubl. data).
Further Resources
The use of photographs by Luke Kemp is acknowledged. Other images by Ryan Tippett.
Striped Caco Cacosternum striatum FitzSimons, 1947
Other Common Names: Gestreepte Blikslanertjie (Afrikaans)
Recommended citation format: Scott, E; Tippett, RM. (2025). Striped Caco Cacosternum striatum. Biodiversity and Development Institute, Cape Town. Available online at https://thebdi.org/2022/02/20/striped-caco-cacosternum-striatum/
This species text has been updated and expanded from the text in the
2004 frog atlas: Scott, E. (2004). Striped Caco Cacosternum striatum. In Minter LR et al 2004.
References:
Minter, LR; Burger, M; Harrison, JA; Braack, HH; Bishop, PJ; Kloepfer, D. (Editors). (2004). Atlas and Red Data Book of the Frogs of South Africa, Lesotho and Swaziland. Smithsonian Institution, Washington, and Avian Demography
Unit, Cape Town.
Carruthers, V; du Preez, L. (2017). Frogs of southern Africa: A Complete Guide. Struik Nature, Cape Town.
Channing, A. (2001) Amphibians of Central and Southern Africa. Protea Book House, Pretoria