Fanie Rautenbach has worked his way through the LepiMAP records for South Africa, Lesotho and Swaziland, has identified data gaps, and is challenging us to fill them. Fanie is the member of LepSoc Africa who takes the main responsibility for doing the identifications of butterflies in LepiMAP.

This blog does two things. Firstly, it lists all the species of butterfly for which we don’t have a photographic record in LepiMAP. Secondly, it lists the species for which there are photographic records in LepiMAP, but for which the most recent record was made more than five years ago, i.e. in 2014 or earlier. The Great LepiMAP Challenge for the summer of 2020/21 is to “refresh” the records for these species.
The LepiMAP database contains 477,000 records of butterflies, getting on for half a million. This is one of the largest databases of its kind in the world. For the butterfly atlas of South Africa, Lesotho and eSwatini (the SABCA project), we computerized every specimen record we could find. This included museums and private collections. This database goes back to the 19th century, although most of the records used for the butterfly atlas were made after the Second World War, in the second half of the 20th century, and from 2000 to 2010.
This blog consists of a series of four tables. Each table covers a time period. The first three tables are the 112 species that are not in LepiMAP as photographic records. The fourth table selects the 56 species that have not been “refreshed”, even once, since 2014. The column headers are mostly self-explanatory; the numbers in the column QDGC give the number of quarter degree grid cells in which the species has been recorded; the column records gives the total number of records for the species in the database. The two-letter codes are abbreviations for the provinces of South Africa, eS is eSwatini (Swaziland) and Les is Lesotho. The crosses show which of these political regions the species has been recorded in. Locality gives the general area where the species has been recorded. The numbers in the first column are in date order.
So Fanie’s first challenge to all LepiMAPpers is to get photographic records for these 168 species, and to upload them into the Virtual Museum. In the tables he has indicated the political regions they occur in, and there is a general description of the locality.
Fanie’s second challenge is to find the species in areas where they have not been recorded previously. (Many species have “traditional” sites where they get recorded, and for some species it is believed that they occur only on a single hilltop.) And then people have unexpectedly bumped into them at other sites, often at a considerable distance. Steve Woodhall’s talk at a BDI Citizen Scientist Hour called “Looking for butterflies” will help you learn how to go about doing butterfly fieldwork. And his book: “Fieldguide to Butterflies of South Africa” (reviewed here) gives the flight period for each species, so you know when to look. You can order a copy of this field guide by sending an email to Steve Woodhall.
We asked Steve to comment on this challenge: “This challenge is vital, as it carries on the great work done by LepSoc Africa and citizen scientists during the original SABCA project. Our understanding of our butterflies’ biogeography was increased immensely by SABCA, but LepiMAP record collecting for the project ended in July 2010, leading up to the publication of the Red List and Atlas in 2013. Environmental change has accelerated in the past 10 years, and we need to rise to this challenge to fill in the new gaps that Fanie has identified.”
This first table lists the 18 species for which there is not a single record in the LepiMAP database this century. The most recent record was made two decades ago. Amazingly, 12 of these 19 species are regarded as “Least Concern”. But this is due to the reality that many of them have wide ranges farther north in Africa. But it would be nice to know that the species persists at the edge of its range, in the southern end of Africa. (Because of the constraints of this format, you need to scroll out to the right to see the far ends of the tables – sorry! You can also download the tables as an Excel spreadsheet, or as a PDF)
| Family | Scientific name | Common name | Status | QDGC | Rec-ords | Last observed | Li | Mp | Ga | NW | KZN | FS | NC | WC | EC | eS | Les | Locality | |
| 1 | LYCAENIDAE | Deloneura immaculata | Bashee river buff | Extinct | 1 | 2 | 29/12/1863 | X | Bashee River near Gwetyibeni (Fort Bowker) | ||||||||||
| 2 | NYMPHALIDAE | Tirumala petiverana | Dappled monarch, blue monarch | Least Concern | 2 | 2 | 26/06/1976 | X | Malta forest (Lekgalameetse NR) | ||||||||||
| 3 | NYMPHALIDAE | Ypthima antennata antennata | Clubbed ringlet | Least Concern | 4 | 21 | 03/02/1979 | X | Buffelsberg near Munnik (NE of Polokwane) | ||||||||||
| 4 | NYMPHALIDAE | Stygionympha dicksoni | Dickson’s hillside brown | Critically Endangered | 3 | 32 | 31/08/1985 | X | Tygerberg, Kapokberg (Darling) | ||||||||||
| 5 | LYCAENIDAE | Aloeides mbuluensis | Mbulu’s copper | Least Concern | 3 | 30 | 20/11/1990 | X | X | Mbulu (Transkei) Lotheni | |||||||||
| 6 | NYMPHALIDAE | Pseudonympha southeyi southeyi | Southey’s brown | Least Concern | 7 | 53 | 10/11/1991 | X | Joubert’s Pass (Lady Grey), Ben MacDhui | ||||||||||
| 7 | HESPERIIDAE | Andronymus caesar philander | White dart | Least Concern | 4 | 6 | 16/05/1993 | X | X | X | Kosi Bay, Pafuri (Kruger park) Swadini resort | ||||||||
| 8 | NYMPHALIDAE | Charaxes druceanus solitarius | Silver-barred charaxes | Least Concern | 1 | 4 | 19/07/1993 | X | Blouberg | ||||||||||
| 9 | LYCAENIDAE | Thestor calviniae | Dickson’s skolly | Least Concern | 1 | 46 | 11/11/1993 | X | mear Calvinia, Hantamsberg | ||||||||||
| 10 | NYMPHALIDAE | Coenyropsis natalii poetulodes | Natal brown | Data Deficient | 3 | 27 | 06/03/1994 | X | Chuniespoort, Tubex (Wolkberg) | ||||||||||
| 11 | NYMPHALIDAE | Charaxes xiphares staudei | Forest-king charaxes | Least Concern | 1 | 16 | 14/03/1994 | X | Blouberg | ||||||||||
| 12 | NYMPHALIDAE | Dira swanepoeli isolata | Swanepoel’s widow | Least Concern | 1 | 23 | 12/02/1996 | X | Blouberg | ||||||||||
| 13 | LYCAENIDAE | Thestor protumnus terblanchei | Terblanche’s skolly | Vulnerable | 2 | 8 | 15/02/1996 | X | Korannaberg | ||||||||||
| 14 | LYCAENIDAE | Trimenia malagrida malagrida | Scarce mountain copper | Critically Endangered | 1 | 120 | 26/02/1996 | X | Lion’s Head, Twelve Apostles | ||||||||||
| 15 | NYMPHALIDAE | Neptis serena serena | Serene sailer | Least Concern | 5 | 6 | 10/09/1996 | X | Near Mpaphuli Cycad Reserve | ||||||||||
| 16 | LYCAENIDAE | Chrysoritis lyncurium | Tsomo river opal | Vulnerable | 4 | 84 | 18/12/1997 | X | Mbulu Forest, Tsomo River | ||||||||||
| 17 | NYMPHALIDAE | Sevenia rosa | Rosa’s tree nymph | Least Concern | 5 | 17 | 19/01/1998 | X | Ngoye Forest Reserve | ||||||||||
| 18 | LYCAENIDAE | Trimenia wykehami | Wykeham’s silver-spotted copper | Least Concern | 8 | 176 | 11/12/1999 | X | X | Verlatenkloof Pass (Sutherland), Nuweveld mountains (Beaufort West) |
The next table contains the 19 butterfly species that were last recorded between 2000 and 2007.
| Family | Scientific name | Common name | Status | QDGC | Rec-ords | Last observed | Li | Mp | Ga | NW | KZN | FS | NC | WC | EC | eS | Les | Locality | |
| 19 | LYCAENIDAE | Aloeides carolynnae aurata | Carolynn’s copper | Near Threatened | 5 | 77 | 21/02/2000 | X | De Hoop Nature Reserve, Still Bay | ||||||||||
| 20 | LYCAENIDAE | Chrysoritis thysbe mithras | Common opal | Data Deficient | 2 | 48 | 01/09/2000 | X | Brenton-on-sea, Struisbaai | ||||||||||
| 21 | LYCAENIDAE | Chrysoritis oreas | Drakensberg daisy copper | Near Threatened | 10 | 197 | 16/10/2000 | X | Bulwer Mountain, Lotheni, Bushman’s Neck” | ||||||||||
| 22 | LYCAENIDAE | Thestor petra tempe | Tempe skolly | Least Concern | 5 | 73 | 29/12/2000 | X | Elandsberg, Seweweekspoort | ||||||||||
| 23 | NYMPHALIDAE | Pseudonympha southeyi kamiesbergensis | Southey’s brown | Least Concern | 5 | 26 | 03/10/2001 | X | Grootvlei pass (Kamieskroon) | ||||||||||
| 24 | HESPERIIDAE | Kedestes sarahae | Sarah’s ranger | Least Concern | 1 | 13 | 14/10/2001 | X | Cedarberg Mountains (Welbedacht) | ||||||||||
| 25 | LYCAENIDAE | Aloeides monticola | Cedarberg copper | Least Concern | 1 | 43 | 21/10/2001 | X | Cedarberg Mountains | ||||||||||
| 26 | LYCAENIDAE | Chrysoritis blencathrae | Waaihoek opal | Least Concern | 2 | 72 | 21/01/2002 | X | Waaihoek Mountains (Worcester) | ||||||||||
| 27 | LYCAENIDAE | Zintha hintza krooni | Hintza pierrot | Least Concern | 4 | 4 | 16/12/2002 | X | Witsand | ||||||||||
| 28 | LYCAENIDAE | Trimenia wallengrenii wallengrenii | Wallengren’s silver-spotted copper | Critically Endangered | 1 | 21 | 17/11/2003 | X | Kapokberg near Darling | ||||||||||
| 29 | NYMPHALIDAE | Torynesis orangica | Orange widow | Least Concern | 4 | 282 | 15/02/2004 | X | Golden gate, Clarens | ||||||||||
| 30 | LYCAENIDAE | Virachola dinomenes dinomenes | Orange playboy | Least Concern | 17 | 240 | 18/06/2004 | X | Hluhluwe False Bay Park | ||||||||||
| 31 | LYCAENIDAE | Aloeides stevensoni | Stevenson’s copper | Endangered | 2 | 64 | 06/11/2004 | X | Haenertsburg | ||||||||||
| 32 | LYCAENIDAE | Chrysoritis adonis adonis | Adonis opal | Least Concern | 2 | 186 | 14/11/2005 | X | Gydo Mountain (Ceres) | ||||||||||
| 33 | NYMPHALIDAE | Neita lotenia | Loteni brown | Least Concern | 7 | 103 | 07/01/2006 | X | X | Bushman’s Neck, Lotheni | |||||||||
| 34 | NYMPHALIDAE | Torynesis pringlei | Pringle’s widow | Least Concern | 4 | 42 | 28/01/2007 | X | Rafoletsane, Sehonghong Valley | ||||||||||
| 35 | NYMPHALIDAE | Cassionympha camdeboo | Camdeboo brown | Least Concern | 3 | 35 | 14/11/2007 | X | Aberdeen | ||||||||||
| 36 | LYCAENIDAE | Lepidochrysops jefferyi | Jeffery’s blue | Endangered | 2 | 160 | 17/11/2007 | X | Barberton | ||||||||||
| 37 | LYCAENIDAE | Lepidochrysops swanepoeli | Swanepoel’s blue | Endangered | 2 | 140 | 17/11/2007 | X | Barberton | ||||||||||
| 38 | LYCAENIDAE | Thestor barbatus | Bearded skolly | Data Deficient | 1 | 13 | 15/12/2007 | X | Spitskoppie (Herold) |
This big table contains the 74 butterfly species which were last recorded between 2008 and 2011, and which are not in LepiMAP as photographic records.
| Family | Scientific name | Common name | Status | QDGC | Rec-ords | Last observed | Li | Mp | Ga | NW | KZN | FS | NC | WC | EC | eS | Les | Locality | |
| 39 | LYCAENIDAE | Lepidochrysops loewensteini | Loewenstein’s blue | Least Concern | 5 | 56 | 13/01/2008 | X | X | Sehonghong river valley Dulcies neck | |||||||||
| 40 | LYCAENIDAE | Orachrysops nasutus remus | Nosy blue | Least Concern | 16 | 84 | 13/01/2008 | X | X | X | Ongeluksnek Sehonghong river valley | ||||||||
| 41 | LYCAENIDAE | Aloeides pringlei | Pringle’s copper | Least Concern | 2 | 77 | 15/02/2008 | X | Groot Winterberg (Between Adelaide and Tarkastad) | ||||||||||
| 42 | LYCAENIDAE | Anthene minima minima | Little hairtail | Least Concern | 25 | 81 | 14/03/2008 | X | X | X | X | Mhlosinga Mkuse | |||||||
| 43 | NYMPHALIDAE | Charaxes marieps | Marieps charaxes | Least Concern | 1 | 121 | 29/03/2008 | X | X | Mariepskop | |||||||||
| 44 | LYCAENIDAE | Lepidochrysops penningtoni | Pennington’s blue | Data Deficient | 4 | 23 | 03/09/2008 | X | Wolfhok | ||||||||||
| 45 | HESPERIIDAE | Spialia agylla bamptoni | Grassveld sandman | Least Concern | 4 | 43 | 21/09/2008 | X | Hondeklip bay | ||||||||||
| 46 | PIERIDAE | Colotis celimene pholoe | Lilac tip | Least Concern | 3 | 6 | 27/09/2008 | X | Tswalu Nature Reserve | ||||||||||
| 47 | LYCAENIDAE | Chrysoritis turneri wykehami | Wykeham’s opal | Least Concern | 3 | 78 | 18/10/2008 | X | Hantamsberg Sutherland | ||||||||||
| 48 | LYCAENIDAE | Chrysoritis nigricans rubescens | Dark opal | Least Concern | 1 | 26 | 05/11/2008 | X | Gamka nature reserve | ||||||||||
| 49 | LYCAENIDAE | Chrysoritis azurius | Azure opal | Least Concern | 11 | 200 | 21/11/2008 | X | Sutherland Roggeveld escarpment | ||||||||||
| 50 | NYMPHALIDAE | Serradinga bowkeri bella | Bowker’s widow | Least Concern | 6 | 10 | 02/12/2008 | X | Compass berg Swaershoek Pass (Cradock) | ||||||||||
| 51 | LYCAENIDAE | Thestor overbergensis | Overberg skolly | Least Concern | 3 | 15 | 08/12/2008 | X | De Hoop nature reserve Cape Agulhas | ||||||||||
| 52 | LYCAENIDAE | Chloroselas mazoensis | Purple gem | Least Concern | 17 | 145 | 21/12/2008 | X | X | X | X | Watervalsrivierpas (Lydenburg) Tembe Elephant park | |||||||
| 53 | NYMPHALIDAE | Neita durbani | D’Urban’s brown | Least Concern | 13 | 114 | 05/01/2009 | X | Bedford Pearston | ||||||||||
| 54 | LYCAENIDAE | Chrysoritis thysbe schloszae | Moorreesburg Common opal | Critically Endangered | 1 | 52 | 01/03/2009 | X | Koringberg (Moorreesburg) | ||||||||||
| 55 | PAPILIONIDAE | Papilio ophidicephalus zuluensis | Emperor swallowtail | Least Concern | 5 | 110 | 10/03/2009 | X | Nkandla Forest | ||||||||||
| 56 | NYMPHALIDAE | Stygionympha geraldi | Gerald’s hillside brown | Least Concern | 11 | 80 | 17/04/2009 | X | Port Nolloth, Hondeklipbaai | ||||||||||
| 57 | LYCAENIDAE | Capys penningtoni | Pennington’s protea | Endangered | 6 | 330 | 26/09/2009 | X | Bulwer mountain | ||||||||||
| 58 | NYMPHALIDAE | Tarsocera cassus outeniqua | Spring widow | Least Concern | 17 | 87 | 10/10/2009 | X | Calitzdorp Anysberg | ||||||||||
| 59 | LYCAENIDAE | Chrysoritis beaufortia charlesi | Beaufort opal | Least Concern | 3 | 132 | 18/10/2009 | X | Sutherland | ||||||||||
| 60 | LYCAENIDAE | Chrysoritis violescens | Violescent opal | Least Concern | 7 | 239 | 18/10/2009 | X | Sutherland | ||||||||||
| 61 | LYCAENIDAE | Lepidochrysops jamesi jamesi | James’s blue | Least Concern | 6 | 111 | 18/10/2009 | X | Sutherland | ||||||||||
| 62 | LYCAENIDAE | Phasis pringlei | Pringle’s arrowhead | Least Concern | 4 | 78 | 18/10/2009 | X | Sutherland | ||||||||||
| 63 | LYCAENIDAE | Aloeides caledoni | Caledon copper | Least Concern | 8 | 27 | 22/10/2009 | X | X | Lootsberg pass Shaw’s pass | |||||||||
| 64 | LYCAENIDAE | Aloeides merces | Wakkerstroom copper | Least Concern | 7 | 49 | 08/11/2009 | X | X | Dirkiesdorp Mhlonganvula | |||||||||
| 65 | NYMPHALIDAE | Dingana alaedeus | Wakkerstroom widow | Near Threatened | 4 | 74 | 08/11/2009 | X | Kastrolnek (Wakkerstroom) | ||||||||||
| 66 | LYCAENIDAE | Aloeides pallida liversidgei | Giant copper | Least Concern | 3 | 29 | 11/11/2009 | X | Baviaanskloof | ||||||||||
| 67 | LYCAENIDAE | Chrysoritis zeuxo cottrelli | Cottrell’s daisy copper | Least Concern | 10 | 76 | 11/11/2009 | X | Gamka Nature Reserve, Millwood NR | ||||||||||
| 68 | LYCAENIDAE | Lepidochrysops quickelbergei | Quickelberge’s blue | Least Concern | 4 | 73 | 21/11/2009 | X | Gydo Mountain (Ceres) | ||||||||||
| 69 | LYCAENIDAE | Thestor camdeboo | Camdeboo skolly | Least Concern | 4 | 48 | 17/12/2009 | X | Cambedoo Mountains (Aberdeen) | ||||||||||
| 70 | LYCAENIDAE | Thestor compassbergae | Compassberg skolly | Least Concern | 3 | 112 | 18/12/2009 | X | Compassberg (Graaf-Reinett) | ||||||||||
| 71 | NYMPHALIDAE | Neita neita | Neita brown | Least Concern | 18 | 211 | 27/12/2009 | X | X | Watervalsrivierpas | |||||||||
| 72 | LYCAENIDAE | Chrysoritis daphne | Daphne’s opal | Least Concern | 1 | 100 | 29/12/2009 | X | Kammanasie mountains (Uniondale) | ||||||||||
| 73 | LYCAENIDAE | Chrysoritis pyramus balli | Ball’s opal | Least Concern | 3 | 110 | 29/12/2009 | X | Kammanasie mountains (Uniondale) | ||||||||||
| 74 | LYCAENIDAE | Orachrysops brinkmani | Brinkman’s blue | Least Concern | 1 | 76 | 29/12/2009 | X | Kammanasie Mountains (Uniondale) | ||||||||||
| 75 | NYMPHALIDAE | Serradinga kammanassiensis | Kammanassie widow | Least Concern | 2 | 75 | 29/12/2009 | X | Kammanasie Mountains (Uniondale) | ||||||||||
| 76 | NYMPHALIDAE | Pseudonympha penningtoni | Pennington’s brown | Least Concern | 26 | 232 | 31/12/2009 | X | X | X | Ben McDhui, Mount-Aux-Sources, Moteng | ||||||||
| 77 | NYMPHALIDAE | Neptis kiriakoffi | Kiriakoff’s sailer | Least Concern | 5 | 8 | 25/02/2010 | X | X | X | Mangusi, Mpaphuli Cycad Reserve, Kowyns Pas | ||||||||
| 78 | NYMPHALIDAE | Charaxes karkloof trimeni | Karkloof charaxes | Least Concern | 6 | 44 | 07/03/2010 | X | Gouna Forest, Hoogekraal Pass | ||||||||||
| 79 | LYCAENIDAE | Trimenia malagrida paarlensis | Scarce mountain copper | Critically Endangered | 1 | 140 | 13/03/2010 | X | Paarl Mountain, Paardeberg | ||||||||||
| 80 | NYMPHALIDAE | Cyrestis camillus sublineata | African porcelain | Least Concern | 5 | 19 | 20/03/2010 | X | Pafuri | ||||||||||
| 83 | PIERIDAE | Dixeia leucophanes | Spotless black-veined white | 2 | 6 | 03/09/2010 | X | Mabelikwe | |||||||||||
| 84 | NYMPHALIDAE | Tarsocera imitator | Deceptive widow | Least Concern | 28 | 147 | 04/09/2010 | X | X | Springbok Wolfhok | |||||||||
| 85 | LYCAENIDAE | Lepidochrysops jamesi claassensi | James’s blue | Least Concern | 2 | 34 | 05/09/2010 | X | Hantambsberg | ||||||||||
| 86 | LYCAENIDAE | Lepidochrysops badhami | Badham’s blue | Least Concern | 10 | 173 | 02/10/2010 | X | Springbok Caroulsberg | ||||||||||
| 87 | LYCAENIDAE | Thestor dryburghi | Dryburg’s skolly | Least Concern | 8 | 135 | 02/10/2010 | X | Arkoop (N7 north of Kammieskroon) Springbok | ||||||||||
| 88 | LYCAENIDAE | Lepidochrysops bacchus | Wineland blue | Least Concern | 23 | 158 | 03/10/2010 | X | X | Malmesbury, Coega (PE) | |||||||||
| 89 | LYCAENIDAE | Chrysoritis uranus schoemani | Uranus opal | Least Concern | 6 | 41 | 09/10/2010 | X | Gifberg | ||||||||||
| 90 | LYCAENIDAE | Orachrysops regalis | Royal blue | Least Concern | 6 | 48 | 09/10/2010 | X | Wolkberg | ||||||||||
| 91 | LYCAENIDAE | Thestor rooibergensis | Rooiberg skolly | Least Concern | 2 | 65 | 16/10/2010 | X | Top of Rooiberg (Calitzdorp) | ||||||||||
| 92 | HESPERIIDAE | Spialia secessus | Wolkberg sandman | Least Concern | 10 | 97 | 06/11/2010 | X | X | X | Munnik, Sheba Mine | ||||||||
| 93 | LYCAENIDAE | Thestor pictus | Langeberg skolly | Least Concern | 4 | 71 | 10/11/2010 | X | Garcia’s Pass | ||||||||||
| 94 | LYCAENIDAE | Aloeides pallida jonathani | Giant copper | Least Concern | 1 | 93 | 19/11/2010 | X | Kammanassie Mountains | ||||||||||
| 95 | LYCAENIDAE | Anthene millari | Millar’s hairtail | Least Concern | 47 | 220 | 19/11/2010 | X | X | X | X | X | X | Various locations | |||||
| 96 | LYCAENIDAE | Lepidochrysops balli | Ball’s blue | Least Concern | 2 | 98 | 19/11/2010 | X | Kammanassie Mountains | ||||||||||
| 97 | LYCAENIDAE | Deloneura millari millari | Millar’s buff | 21 | 218 | 21/11/2010 | X | X | Makatini Flats, Emanguzi Forest | ||||||||||
| 98 | LYCAENIDAE | Chrysoritis adonis aridimontis | Adonis opal | Least Concern | 1 | 67 | 08/12/2010 | X | Elandsberg | ||||||||||
| 99 | LYCAENIDAE | Lepidochrysops littoralis | Coastal blue | Near Threatened | 14 | 129 | 12/12/2010 | X | Witsand Stillbay, Mosselbay | ||||||||||
| 100 | LYCAENIDAE | Lepidochrysops victori | Victor’s blue | Vulnerable | 2 | 53 | 20/12/2010 | X | Huntley Glen, Long hill (Queenstown) | ||||||||||
| 101 | LYCAENIDAE | Chrysoritis penningtoni | Pennington’s opal | Vulnerable | 6 | 161 | 21/12/2010 | X | Hogsback Mount Kubusi (Stutterheim) | ||||||||||
| 102 | LYCAENIDAE | Orachrysops nasutus nasutus | Nosy blue | Least Concern | 17 | 101 | 21/12/2010 | X | Lundean’s Neck (Barkley East), Gaika’s Kop | ||||||||||
| 103 | LYCAENIDAE | Aloeides dentatis maseruna | Roodepoort copper | Least Concern | 24 | 145 | 22/12/2010 | X | Heilbron | ||||||||||
| 104 | LYCAENIDAE | Aloeides trimeni southeyae | Trimen’s copper | Endangered | 5 | 64 | 22/12/2010 | X | Albertinia | ||||||||||
| 105 | LYCAENIDAE | Aslauga australis | Southern purple | Near Threatened | 10 | 12 | 22/12/2010 | X | Tsomo, Cintsa Bay | ||||||||||
| 106 | LYCAENIDAE | Lepidochrysops oosthuizeni | Oosthuizen’s blue | Least Concern | 17 | 106 | 23/12/2010 | X | X | X | Clarens Joubert’s Pass, Lundean’s Neck, Sterkspruit | ||||||||
| 107 | LYCAENIDAE | Lepidochrysops southeyae | Southey’s blue | Least Concern | 15 | 125 | 23/12/2010 | X | X | Sterkspruit, Tarkastad | |||||||||
| 108 | LYCAENIDAE | Lepidochrysops rossouwi | Rossouw’s blue | Least Concern | 3 | 90 | 08/01/2011 | X | Stoffberg | ||||||||||
| 109 | LYCAENIDAE | Lepidochrysops outeniqua | Outeniqua blue | Least Concern | 5 | 79 | 19/01/2011 | X | X | Tsitsikama National Park, Avontuur | |||||||||
| 110 | LYCAENIDAE | Lepidochrysops vansoni | Van Son’s blue | Least Concern | 12 | 67 | 12/02/2011 | X | X | Loding, Kopje, Great Saltpan | |||||||||
| 111 | LYCAENIDAE | Trimenia malagrida maryae | Scarce mountain copper | Least Concern | 2 | 110 | 22/02/2011 | X | Near Vermaaklikheid | ||||||||||
| 118 | LYCAENIDAE | Lepidochrysops hypopolia | Morant’s blue | Extinct | 1 | 1 | 26/10/2011 | X | X | Blue bank (Ladysmith), Potchefstroom | |||||||||
| 119 | LYCAENIDAE | Chrysoritis felthami dukei | Feltham’s opal | Least Concern | 28 | 127 | 29/10/2011 | X | X | Worcester/Robertson area, Sutherland | |||||||||
| 120 | NYMPHALIDAE | Dingana clara | Clara’s widow | Endangered | 4 | 68 | 30/10/2011 | X | Wolkberg |
The table below the map & photo contains 56 species for which there are photographic records in LepiMAP, but the most recent record is five or more years old, and needs to be refreshed. The map and photo below are representative of this group of species. There are 56 records in the LepiMAP database going back to 1915. They come from 25 scattered quarter degree grid cells. But this species has eluded LepiMAPpers since Vaughan Jessnitz took this photo near Hoedspruit in 2011!
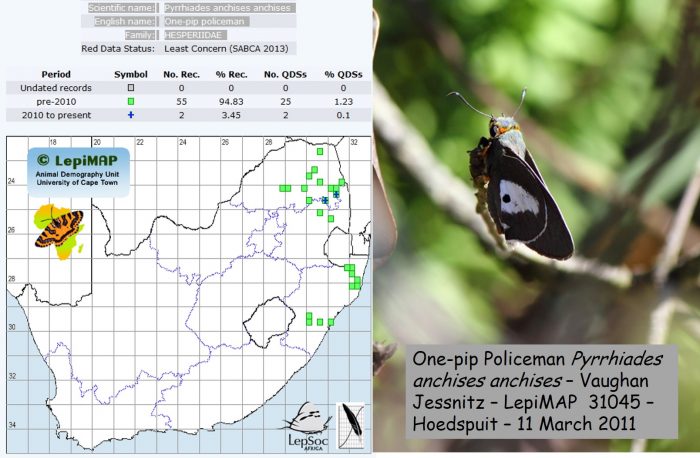
For a species to qualify for the table below, it needs to have a photographic record in LepiMAP. The cut-off date for being eligible is that the most recent record in LepiMAP must have been before 31 December 2014.
| Family | Scientific name | Common name | Status | QDGC | Rec-ords | Last observed | Li | Mp | Ga | NW | KZN | FS | NC | WC | EC | eS | Les | Locality | |
| 112 | HESPERIIDAE | Pyrrhiades anchises anchises | One-pip policeman | Least Concern | 25 | 56 | 11/03/2011 | X | X | X | Imfolozi Nature Reserve, Linwood forest, Klaserie private NR | ||||||||
| 113 | NYMPHALIDAE | Ypthima condamini condamini | Condamin’s ringlet | Least Concern | 2 | 2 | 15/03/2011 | X | Wolkberg farm at Letaba drift | ||||||||||
| 114 | NYMPHALIDAE | Precis antilope | Darker commodore | Least Concern | 40 | 72 | 22/04/2011 | X | X | X | X | Nylsvley Nature Reserve, Mlawula Nature Reserve | |||||||
| 115 | LYCAENIDAE | Lepidochrysops wykehami | Wykeham’s blue | Least Concern | 7 | 149 | 19/09/2011 | X | Kamieskroon, Wolfhok | ||||||||||
| 116 | NYMPHALIDAE | Pseudonympha trimenii nieuwveldensis | Trimen’s brown | Least Concern | 5 | 68 | 19/09/2011 | X | Molteno pass (Nuweveld mountains) | ||||||||||
| 117 | LYCAENIDAE | Aloeides kaplani | Kaplan’s copper | Least Concern | 13 | 91 | 20/09/2011 | X | X | Sutherland, Karoo National Park (Beaufort West) | |||||||||
| 121 | NYMPHALIDAE | Serradinga clarki ocra | Clark’s widow | Least Concern | 2 | 43 | 04/11/2011 | X | Long Tom pass | ||||||||||
| 122 | LYCAENIDAE | Aloeides barbarae | Barbara’s copper | Endangered | 2 | 45 | 11/11/2011 | X | Barberton | ||||||||||
| 123 | LYCAENIDAE | Iolaus lulua | White spotted sapphire | Least Concern | 5 | 50 | 07/12/2011 | X | Ndumu game reserve | ||||||||||
| 124 | NYMPHALIDAE | Pseudonympha paragaika | Golden Gate brown | Vulnerable | 5 | 148 | 02/01/2012 | X | Golden gate | ||||||||||
| 125 | LYCAENIDAE | Lepidochrysops lerothodi | Lesotho blue | Least Concern | 16 | 202 | 21/01/2012 | X | X | Mokhotlong, Blue mountain pass, Lundean’s Nek | |||||||||
| 126 | NYMPHALIDAE | Charaxes xiphares xiphares | Forest-king charaxes | Least Concern | 12 | 98 | 19/03/2012 | X | X | Tsitsikama National Park, Saarsveld, Karatara | |||||||||
| 127 | NYMPHALIDAE | Charaxes xiphares bavenda | Forest-king charaxes | Least Concern | 10 | 118 | 29/03/2012 | X | Hangklip forest, Entabeni forest | ||||||||||
| 128 | LYCAENIDAE | Durbania limbata | Natal rocksitter | Least Concern | 23 | 426 | 21/04/2012 | X | X | Balgowan, Mooirivier, Harrismith | |||||||||
| 129 | LYCAENIDAE | Chrysoritis beaufortia stepheni | Stephen’s opal | Least Concern | 5 | 351 | 06/10/2012 | X | Hantamsberg, Wolfhok | ||||||||||
| 130 | LYCAENIDAE | Chrysoritis phosphor borealis | Scarce scarlet | Least Concern | 10 | 75 | 06/10/2012 | X | X | Panorama gorge, Mhlopeni Nature Reserve | |||||||||
| 131 | LYCAENIDAE | Aloeides simplex | Dune copper | Least Concern | 19 | 139 | 01/11/2012 | X | Witsand, Kuruman | ||||||||||
| 132 | NYMPHALIDAE | Charaxes pondoensis | Pondo charaxes | Least Concern | 13 | 159 | 04/01/2013 | X | X | Port St. Johns, Umtamvuma reserve | |||||||||
| 133 | LYCAENIDAE | Alaena margaritacea | Wolkberg zulu | Critically Endangered | 2 | 259 | 05/01/2013 | X | Wolkberg | ||||||||||
| 134 | LYCAENIDAE | Lepidochrysops braueri | Brauer’s blue | Least Concern | 21 | 153 | 08/01/2013 | X | X | Prince Alfred’s pass, Buffelsnek, Baviaanskloof | |||||||||
| 135 | NYMPHALIDAE | Stygionympha vansoni | Van Son’s hillside brown | Least Concern | 17 | 158 | 24/04/2013 | X | Springbok, Kamieskroon, Carolusberg | ||||||||||
| 136 | LYCAENIDAE | Anthene talboti | Talbot’s hairtail | Least Concern | 73 | 169 | 14/06/2013 | X | X | X | X | X | X | X | X | X | Various locations | ||
| 137 | LYCAENIDAE | Chrysoritis lyndseyae | Lyndsey’s Opal | Vulnerable | 1 | 1 | 25/09/2013 | X | Hondeklipbaai (Wallekraal road) | ||||||||||
| 138 | LYCAENIDAE | Orachrysops mijburghi | Mijburgh’s blue | Endangered | 5 | 72 | 27/10/2013 | X | X | Heilbron, Suikerbosrand | |||||||||
| 139 | LYCAENIDAE | Thestor vansoni | Van Son’s skolly | Least Concern | 3 | 114 | 02/11/2013 | X | Gydo mountain (Ceres) | ||||||||||
| 140 | HESPERIIDAE | Platylesches dolomitica | Hilltop hopper | Least Concern | 10 | 19 | 16/11/2013 | X | X | Utopia nature reserve, Hillshaven | |||||||||
| 141 | LYCAENIDAE | Aloeides molomo krooni | Molomo copper | Least Concern | 13 | 93 | 18/11/2013 | X | Kuruman, Witsand | ||||||||||
| 142 | LYCAENIDAE | Trimenia wallengrenii gonnemoi | Wallengren’s silver-spotted copper | Vulnerable | 3 | 32 | 18/11/2013 | X | Piketberg | ||||||||||
| 143 | LYCAENIDAE | Erikssonia edgei | Erikson’s copper | Critically Endangered | 2 | 61 | 14/12/2013 | X | Bateleur nature reserve | ||||||||||
| 144 | LYCAENIDAE | Orachrysops montanus | Golden Gate blue | Least Concern | 3 | 45 | 18/12/2013 | X | Golden gate | ||||||||||
| 145 | LYCAENIDAE | Aloeides bamptoni | Bampton’s copper | Least Concern | 27 | 172 | 09/01/2014 | X | Springbok, Witsand, Steinkopf | ||||||||||
| 146 | PAPILIONIDAE | Papilio ophidicephalus ayresi | Emperor swallowtail | Least Concern | 15 | 138 | 31/01/2014 | X | X | X | Mariepskop, Nongoma Ngome Forest | ||||||||
| 147 | HESPERIIDAE | Spialia depauperata australis | Wandering sandman | Least Concern | 48 | 211 | 01/02/2014 | X | X | X | X | X | Utopia nature reserve, Tswaing Nature Reserve | ||||||
| 148 | HESPERIIDAE | Platylesches tina | Small hopper | Least Concern | 6 | 13 | 15/02/2014 | X | Lekgalameetse Nature Reserve | ||||||||||
| 149 | LYCAENIDAE | Chrysoritis aureus | Heidelberg opal | Endangered | 5 | 215 | 08/03/2014 | X | X | Alice Glockner Reserve | |||||||||
| 150 | NYMPHALIDAE | Neita extensa | Savanna brown | Least Concern | 35 | 193 | 08/03/2014 | X | X | Perdekop (Jan Trichardt pass), Verloren vallei (Dullstroom) | |||||||||
| 151 | LYCAENIDAE | Thestor dicksoni warreni | Dickson’s skolly | Data Deficient | 2 | 45 | 05/04/2014 | X | Graafwater | ||||||||||
| 152 | NYMPHALIDAE | Cymothoe alcimeda marieps | Battling glider | Least Concern | 17 | 143 | 12/04/2014 | X | Mariepskop | ||||||||||
| 153 | NYMPHALIDAE | Torynesis mintha piquetbergensis | Mintha widow | Least Concern | 4 | 123 | 07/05/2014 | X | Hill NE of Moorreesburg | ||||||||||
| 154 | PIERIDAE | Colotis celimene amina | Lilac tip | Least Concern | 32 | 191 | 29/05/2014 | X | X | X | X | X | Various locations | ||||||
| 155 | HESPERIIDAE | Teniorhinus harona | Arrowhead orange | 2 | 2 | 14/06/2014 | X | Mphapuli Cycad Reserve | |||||||||||
| 156 | LYCAENIDAE | Chrysoritis dicksoni | Dickson’s Strandveld copper | Critically Endangered | 1 | 49 | 09/09/2014 | X | Witsand | ||||||||||
| 157 | LYCAENIDAE | Chrysoritis zonarius coetzeri | Coetzer’s daisy copper | Least Concern | 7 | 96 | 12/09/2014 | X | X | Nieuwoudtville, Clanwilliam | |||||||||
| 158 | NYMPHALIDAE | Telchinia induna salmontana | Induna acraea | Endangered | 7 | 62 | 13/09/2014 | X | Lajuma mountain retreat, Matshavhawe | ||||||||||
| 159 | LYCAENIDAE | Lepidochrysops mcgregori | McGregor’s blue | Least Concern | 4 | 128 | 19/09/2014 | X | Hantam national botanical garden | ||||||||||
| 160 | LYCAENIDAE | Chrysoritis thysbe psyche | Common opal | Least Concern | 6 | 189 | 20/09/2014 | X | Bitterfontein, Lambert’s bay | ||||||||||
| 161 | NYMPHALIDAE | Stygionympha scotina coetzeri | Eastern hillside brown | Least Concern | 6 | 11 | 25/09/2014 | X | X | Lekgalameetse Nature Reserve | |||||||||
| 162 | PIERIDAE | Colotis doubledayi | Desert veined tip | Least Concern | 6 | 173 | 25/09/2014 | X | Vioolsdrift | ||||||||||
| 163 | LYCAENIDAE | Trimenia argyroplaga cardouwae | Large silver-spotted copper | Least Concern | 4 | 34 | 21/11/2014 | X | Dasklip pass (Porterville) | ||||||||||
| 164 | NYMPHALIDAE | Pseudonympha swanepoeli | Swanepoel’s brown | Data Deficient | 4 | 43 | 26/11/2014 | X | X | Verlorenvallei, Houtbosberg | |||||||||
| 165 | LYCAENIDAE | Lepidochrysops gydoae | Gydo blue | Least Concern | 1 | 87 | 05/12/2014 | X | Gydo mountain (Ceres) | ||||||||||
| 166 | LYCAENIDAE | Thestor petra petra | Rock skolly | Least Concern | 4 | 245 | 05/12/2014 | X | Gydo mountain (Ceres), Matroosberg | ||||||||||
| 167 | LYCAENIDAE | Chrysoritis turneri amatola | Turner’s opal | Least Concern | 12 | 97 | 14/12/2014 | X | X | Gaika’s kop (Hogsback), Lootsberg pass, Mount Kubusie, Groot Winterberg | |||||||||
| 168 | LYCAENIDAE | Lepidochrysops procera | Potchefstroom blue | Least Concern | 21 | 181 | 20/12/2014 | X | X | X | X | Badplaas, Welverdiend (Gauteng), Hillshaven (Gauteng) |
These 56 species have eluded LepiMAPpers since the beginning of 2015. Let us (a) refresh them, and (b) find new localities where they occur. Ten of the species are in threat categories. Two are “Data Deficient” (we don’t have enough information to classify them into a threat category). The rest are “Least Concern” (we have no worries about the persistence into the future). The way to confirm this “Least Concern” status is to get a better understanding of their distributions, and to maintain a steady stream of records, so we are certain that they are still present.

Although this blog focuses on the southern end of Africa, LepiMAP covers the whole continent, and the offshore islands. Within a few years, it would be fantastic to have sufficient data to do a blog like this for other countries and regions of Africa.
Footnote
The Seventh BDI Citizen Scientist Hour, on 9 September 2020, was devoted to butterflies and LepiMAP. Fanie did a presentation on the Great LepiMAP Challenge. You can watch it here:
There were two other presentations on butterflies during this Citizen Scientist Hour. Oskar Brattström’s title was Butterflies – Africa’s most exciting animals and you can watch it here:
And the third presenter was Steve Woodhall, talking about Looking for butterflies. Here it is:
The BDI Citizen Scientist Hours are Zoom events. The presentations are recorded, and are uploaded to the BDI’s YouTube channel, which is here. To find out when the next Citizen Scientist Hour will take place, go to the Facebook page of BDI Citizen Science, and search under “events”.












































