View the above photo record (by Jennifer Sterne) in FrogMAP here.
Find the Guttural Toad in the FBIS database (Freshwater Biodiversity Information System) here.
Family Bufonidae
GUTTURAL TOAD – Sclerophrys gutturalis
(Power, 1927)
Habitat
B. gutturalis inhabits various vegetation types in the Savanna, Grassland and Thicket biomes at altitudes ranging from sea-level to about 1800 m. In the east, the species is sometimes found in forest clearings and forest/grassland ecotones, while in the west it has a linear distribution along the wooded banks of the Gariep River.
These toads are opportunistic in their selection of sites for aestivation during winter. Quiescent toads have been found in termitaria and in the burrows of a large lizard, the Sungazer lizard Cordylus giganteus (Du Preez 1996).
Breeding takes place in open, shallow pools in rivers, farm dams, garden ponds, canals, ditches and borrow pits. During the day, the frogs take shelter under logs, rocks or other objects, in drain-pipes and gutters, in burrows, or in holes that they excavate in soft ground.
Behaviour
B. gutturalis is active from August to April, and occasionally during warm periods in winter. Breeding begins in small permanent water bodies, such as garden ponds, as soon as temperatures start to rise in spring, often before the first rains arrive. In areas where permanent water bodies do not exist, breeding is initiated by the first heavy spring rains. Dense choruses form, and the rolling, guttural snores of males can be heard from just after dark until well past midnight. Choruses may persist for several weeks, with a peak in late September to November, reviving after every rain shower until mid-February.

Photo by J.K. Boyce
These toads usually call from partly concealed sites, although exposed sites are also used and calling males often seem oblivious to the presence of humans. Calling males exhibit site fidelity, with some individuals returning to the same site year after year. As many as 25 000 eggs, 1.4–1.5 mm diameter, are laid in two gelatinous strings, each 5 mm thick (Wager 1986). Strings of eggs are often twined around aquatic vegetation. Metamorphosis is complete after 5–6 weeks. As soon as the toadlets have developed front legs, they leave the water to hide on the bank until their tails are absorbed, and then start feeding on tiny insects. The maximum longevity, recorded in captivity, is seven years (Channing 2001).
At night they emerge from their shelters to prey on a wide range of insects, spiders and centipedes. In captivity, even lizards and other frogs have been recorded as prey items (Rose 1962). Individuals are often seen feeding on insects attracted to garden lights.
Adults are preyed on by the Black-necked Spitting Cobra Naja nigricollis, Common Night Adder Causus rhombeatus, Western Green Snake Philothamnus angolensis, Serrated Hinged Terrapin Pelusios sinuatus, and African Civet Viverra civetta (Channing 2001). The tadpoles are eaten by aquatic birds, fish and the Common Platanna Xenopus laevis.

Photo by Rob Dickinson
Status and Conservation
B. gutturalis is relatively secure as it is widely distributed, locally abundant and highly adaptable to human settlement. It has been recorded in a number of national parks, provincial nature reserves and other protected areas. However, many individuals are killed by motor vehicles while crossing roads at night, especially during rain showers. On occasion, the toads are needlessly killed by people who are annoyed by their loud calls. The Eastern Cape lineage has a restricted distribution and further surveys are required in this area. Hybridization with B. rangeri occurs at scattered localities and may occur more frequently as a result of modern agricultural practices, but this does not seem to erode the differences between these species and is therefore not considered to be a threat.

Photo by M. Galpin
Distribution
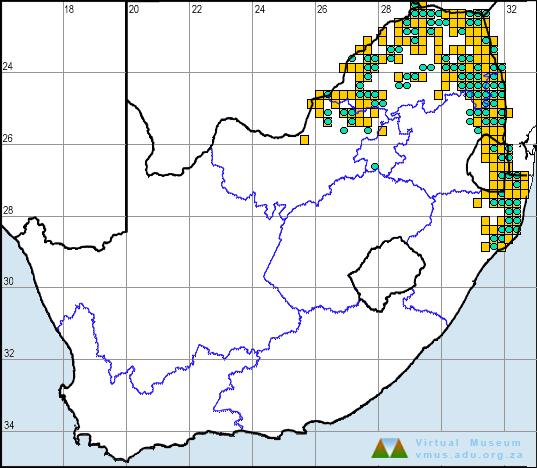
B. gutturalis is distributed from western Uganda eastward to coastal Kenya and southward through Tanzania, Zambia, Malawi, Mozambique, Zimbabwe and Botswana, to South Africa (Tandy 1972). In the atlas region, its distribution is centred in the northeast, particularly in the KwaZulu-Natal, Mpumalanga, Gauteng, central Limpopo, eastern North West, and northern and eastern Free State provinces, and Swaziland. In the Northern Cape Province, its distribution extends westward along the Gariep (Orange) River as far as Goodhouse (2818CC). The species also occurs in Lesotho and northern Eastern Cape Province, but is absent from Western Cape Province except for an introduced population that became established on the Cape Peninsula (3418AB) in recent years.
Earlier workers, including Hewitt (1935, 1937), Poynton (1964), Guttman (1967) and Tandy (1972), recorded B. gutturalis as far south as Port St Johns (3129DA) in Eastern Cape Province. However, during the atlas period several more southerly populations were found, for example, at Hluleka Nature Reserve and surroundings (3129CC, CD), Cwebe Nature Reserve (3228BB), Amalinda Fish Station (3227DD) and Cintsa district (3228CC) near East London. This suggests that the species is expanding its range southward along the east coast. Alternatively, this may simply be the result of a more systematic survey than in the past.
Recent genetic studies of mitochondrial DNA have identified three geographically distinct lineages of B. gutturalis in the atlas region, with additional lineages further north (M.C. pers. obs). One lineage occurs near the Botswana border in the Gaborone district (2425DA), but the extent of this lineage south and west of this area is unknown. A second independent lineage occupies the central area of the range, including KwaZulu-Natal and Mpumulanga provinces and Swaziland. An introduced population in Mauritius, established in the 1940s, originated from this second lineage. The third lineage in the atlas region is restricted to the Eastern Cape populations, and meets the second lineage at Port St Johns. Toads in this southeastern population are smaller than those elsewhere, which led Hewitt (1935) to suggest that they represent a separate subspecies.
B. gutturalis and B. rangeri hybridize at several localities. The hybrids can be identified by aberrant morphology, genetic analysis, and by the fact that their advertisement calls are intermediate in structure to those of their parents. Hybrids have been found at Groenkloof (2528CA), Weza (3029DB) and Port St Johns (3129DA; Guttman 1967; Passmore 1972; M. Cherry in litt.), while an additional four hybrid populations were discovered during atlas surveys at Harding (3029DA), Pietermaritzburg (2930CB), Jamestown (3027CC) and Lubuyane Falls, Swaziland (2631AA).
The absence of records of B. gutturalis in parts of Northern Cape Province and Lesotho is probably due to inaccessibility of these areas rather than to real gaps in distribution. Apart from these limitations, the atlas data are reliable and reasonably comprehensive.

Further Resources
Virtual Museum (FrogMAP > Search VM > By Scientific or Common Name)
More common names: Gorrelskurwepadda (Afrikaans)
Recommended citation format for this species text:
du Preez LH, Weldon C, Cunningham M, Turner A, Tippett RM. Guttural Toad Sclerophrys gutturalis. BDI, Cape Town.
Available online at http://thebdi.org/2021/11/25/guttural-toad-sclerophrys-gutturalis/
Recommended citation format:
This species text has been updated and expanded from the text in the
2004 frog atlas. The reference to the text and the book are as follows:
du Preez LH, Weldon C, Cunningham M, Turner A 2004 Sclerophrys gutturalis Guttural Toad. In Minter LR et al 2004.
Minter LR, Burger M, Harrison JA, Braack HH, Bishop PJ, Kloepfer D (eds)
2004. Atlas and Red Data Book of the Frogs of South Africa, Lesotho and
Swaziland. Smithsonian Institution, Washington, and Avian Demography
Unit, Cape Town.