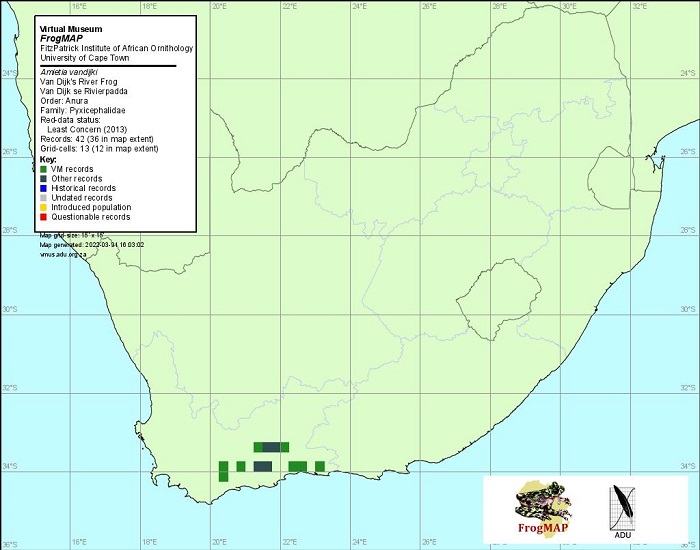
View the above photo record (by John Davies) in FrogMAP here.
Find the Maluti River Frog in the FBIS database (Freshwater Biodiversity Information System) here.
Family Pyxicephalidae
MALUTI RIVER FROG – Amietia vertebralis
(Hewitt, 1927)
Habitat
This species inhabits Afro Montane Grassland and Alti Mountain Grassland of the Grassland Biome, usually at altitudes of 1750–3282 m. It has also been recorded at lower elevations of 1646 m in the Mzimkulu River (2929CB) in Drakensberg Gardens (Bush 1952), and 1540 m at Fort Hook (3027DA) in the Witteberg range (Poynton 1964). These frogs occur in cold, clear streams and associated pools with rocky substrates, in mountainous areas, including most major rivers and their tributaries in Lesotho.

Photo by Michael Cunningham
Behaviour
A. vertebralis is largely aquatic, but may be found on rocks and under stones or amongst vegetation at the water’s edge. Wager (1986) also found adults, up to 75 mm in length, sheltering under stones or water plants in swampy areas on mountain slopes. Adults can apparently remain submerged for a few days at a time (Bush 1952) because they are able to breathe efficiently through their skin, whereas juveniles spend more time foraging at the edge of the water (Lambiris 1988b). Both adults and tadpoles have been observed moving about under ice during winter and are intolerant of high temperatures (Rose 1950; Lambiris 1987, 1989a). Mass mortalities of this species in the Drakensberg (O’Grady 1998) may have been caused by a rise in water temperature in stagnant streams during drought conditions.
Breeding occurs from mid-September to February (Lambiris 1988b). Males usually call while almost completely submerged, with only the head protruding from the water, but occasionally they call from exposed rocks (Channing 1979).
The eggs are attached to submerged vegetation in shallow, gently-flowing streams or associated pools with sandy or stony substrates (Lambiris 1988b, 1989a). Van Dijk (1996) observed eggs in shallow, slow-flowing water in September and late November in central Lesotho. The eggs, c.1.8 mm in diameter, formed a single layer on the substrate and were part of a fairly coherent mass c.30 cm across. Bourquin and Channing (1980) observed egg masses in January, while FitzSimons (1948) collected an egg mass in November that was “attached to vegetable matter in a small backwater, away from the running stream”, and contained larvae “almost ready to emerge”.
Development of tadpoles takes several months or may even exceed a year in the wild (Van Dijk 1996). According to Lambiris (1988b), development in captivity took almost two years.
Tadpoles were found in February in shallow water or along the margins of fast-flowing rivers with rocky substrates (Van Dijk 1996). The tadpole is adapted to life in fast-flowing water in having a wide, flat body, a heavy but narrowly-webbed tail, and a large sucker-like mouth for obtaining a firm grip on smooth rocks. Tadpoles initially prey on infusoria, and later scavenge on detritus (Lambiris 1988b).
Adults prey on invertebrates such as gastropods and crabs, but also consume smaller frogs (Channing 1979). Captive specimens located and devoured small crabs (carapace 30 mm wide) while underwater (Channing 1979), and also ate mice (Rose 1962).

Photo by Cornelia Rautenbach
Status and Conservation
A. vertebralis was previously classified Restricted (Branch 1988). It was subsequently discovered at a number of additional localities, especially in the highlands of Lesotho (Bates 2002), and it appears to be abundant in many parts of its range (e.g. Lambiris 1988b). The species was assessed for this publication and assigned to the category Least Concern because its extent of occurrence and area of occupancy are in excess of the relevant thresholds and, although there are known localized threats to the species, there is no evidence that these are likely to cause a major long-term decline of the species.
The remoteness of this frog’s habitat will undoubtedly protect it from human impacts in most parts of its range. However, A. vertebralis appears to be threatened in areas affected by the Lesotho Highlands Water Project. The filling of the Katse Dam in Lesotho may have isolated some populations (Van Dijk 1996), while filling of the Mohale Dam will result in separation or extinction of populations in the Jorodane, Senqunyane and Bokong rivers (Mouton 1996). If the Mohale Dam causes large-scale migration of people and their livestock to areas further up the river catchment, increased erosion and water pollution will result. This may pose a problem as the species appears to occur only in pristine habitat. These frogs are not known to move overland, thus re-colonization will be prevented by the dam which will act as a barrier to dispersal (Mouton 1996). Littering of stream banks was also identified as a negative factor by Lambiris (1988b), but G. Kopij observed this species in streams polluted by the washing of laundry (Ambrose 1999).
In 1998, the mass mortality of a species assumed to be Amietia vertebralis was reported by hikers at an upland site in the Drakensberg escarpment (O’Grady 1998). The cause of death was undetermined, but at the time the river had been reduced to a series of isolated stagnant pools owing to an exceptionally dry year. The causative organism of chytridiomycosis has been identified in museum specimens of Ametia vertebralis (Speare 2000; Weldon 2002). Although this does not prove that the chytrid fungus was responsible for the deaths reported in 1998, this seems a plausible explanation as chytridiomycosis epidemics often occur during periods of drought, and affect high-altitude species (Berger et al. 1998; Pounds et al. 1999; Berger et al. 2000).

In an opinion survey of local people in the highlands of central Lesotho, it was found that A. vertebralis is probably not threatened by local use as a source of food or medicine. Less than 1% of respondents reported that frogs (as a group) were eaten, while 12% said that they were used for medicine: for example, the fat is used to treat burns (Mouton 1996).
A. vertebralis is protected in Sehlabathebe National Park in Lesotho, Ukhahlamba Drakensberg Mountain Park (World Heritage Site) and by Cape Provincial nature conservation legislation (Nature Conservation Ordinance No. 19 of 1974).
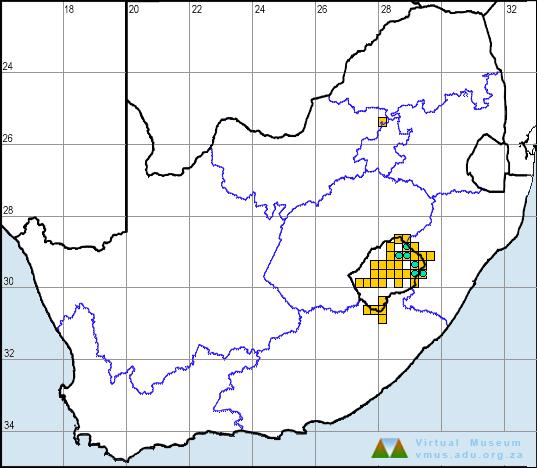
Distribution
A. vertebralis is found at high altitudes in Lesotho and adjacent mountain ranges in KwaZulu-Natal, Free State and Eastern Cape Province, including the Witteberg range (Bates 2002).
Hewitt’s (1927) original description of Rana (now Amietia) vertebralis was based on juvenile frogs. Bush (1952) later distinguished Rana umbraculata from R. vertebralis based on the former’s larger size, relatively wider head and dissimilar sternal apparatus. However, Poynton (1964) presented evidence indicating that R. umbraculata simply represented the adult form of R. vertebralis. Van Dijk (1966) noted morphological differences in tadpole populations that suggested two taxa were represented. He provisionally assigned the tadpoles to R. vertebralis and R. umbraculata. Lambiris (1991) found differences in laryngeal morphology and tadpole mouthparts and also felt that R. vertebralis comprised more than one species.
At the present time only one species of Amietia is recognized, pending further taxonomic research. All atlas distribution data have therefore been referred to A. vertebralis. Bearing in mind the taxonomic uncertainty outlined above, the atlas data may be considered reliable but incomplete.
Further Resources
Virtual Museum (FrogMAP > Search VM > By Scientific or Common Name)
More common names: Aquatic River Frog, Water Rana, Umbraculate Frog, (Alternative English Names); Grootbekrivierpadda (Afrikaans)
Recommended citation format for this species text:
Bates MF, Tippett RM. Maluti River Frog Amietia vertebralis. BDI, Cape Town.
Available online at http://thebdi.org/2022/03/28/maluti-river-frog-amietia-vertebralis/
Recommended citation format:
This species text has been updated and expanded from the text in the
2004 frog atlas. The reference to the text and the book are as follows:
Bates MF 2004 Amietia vertebralis Maluti River Frog. In Minter LR
et al 2004.
Minter LR, Burger M, Harrison JA, Braack HH, Bishop PJ, Kloepfer D (eds)
2004. Atlas and Red Data Book of the Frogs of South Africa, Lesotho and
Swaziland. Smithsonian Institution, Washington, and Avian Demography
Unit, Cape Town.