Cover image of Scaly-feathered Finch by Kyle Finn – Kuruman River, Northern Cape
The Scaly-feathered Finch is a member of the Family PLOCEIDAE (Weavers and Allies). Other members of Ploceidae include the Weavers, Bishops, Widowbirds, & Malimbes. Birds in this family are small passerines. These birds have short to medium, conical, pointed bills, adapted for seed-eating. They also have relatively strong legs and feet. Most are gregarious and are known for their intricately woven nests. They are distributed throughout sub-Saharan Africa, Madagascar, the southern edge of the arabian peninsula, and into tropical Asia. The family Ploceidae contains 123 species from 16 genera. Most species are found in Africa.
Identification
The Scaly-feathered Finch is distinct and unmistakable, and the sexes are alike.

Mapungubwe National Park, Limpopo
Photo by Joanne Putter
The white-tipped black feathers on the crown, forehead, and wings, together with the black malar stripes and rose-pink bill are diagnostic. The remainder of the upper sides are brownish-grey. The undersides, from the throat to the vent, are white. The tail is black with white edges, and the legs and feet are pink.
Immature or juvenile birds resemble adults but lack the scaly-feathering on the forehead and crown, and they also have greyish-yellow (not pink) bills.

Khomas, Namibia
Photo by Norman Barrett
Status and Distribution
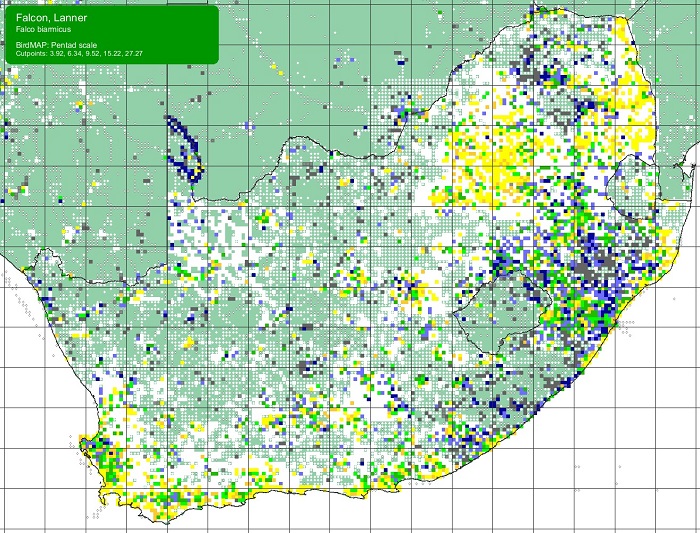
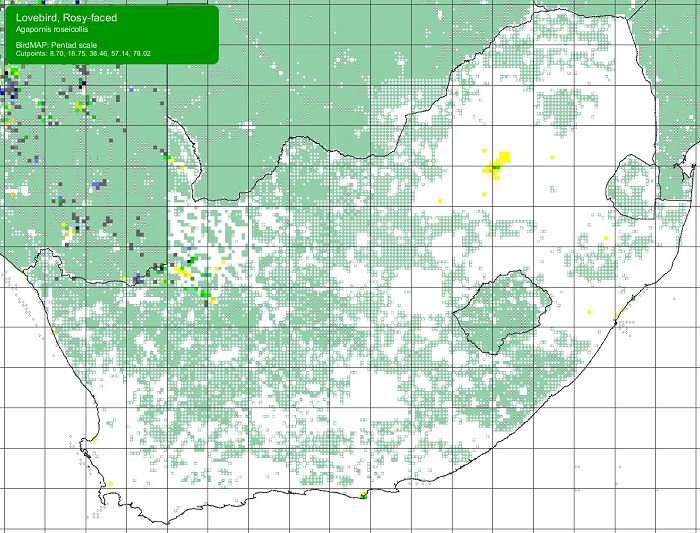
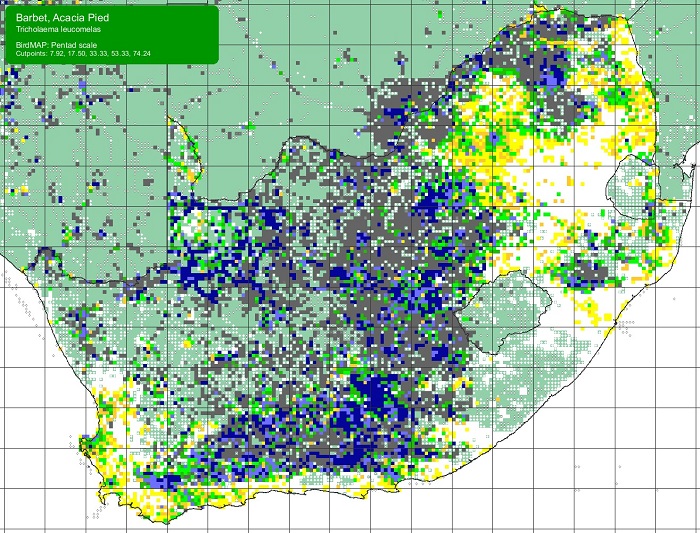
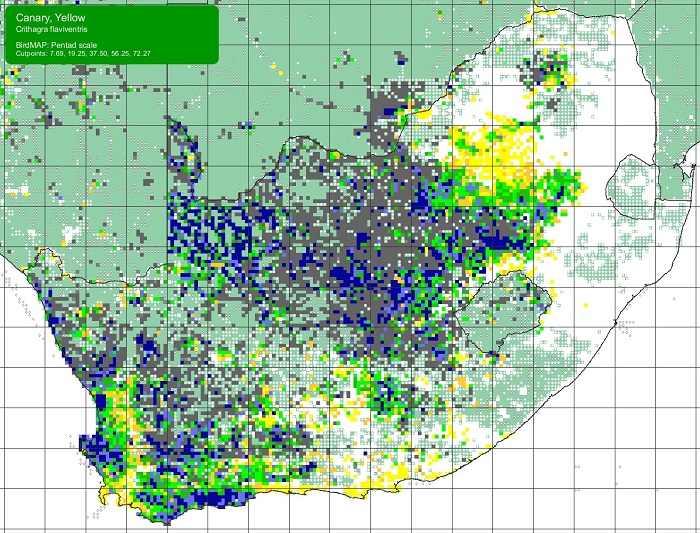
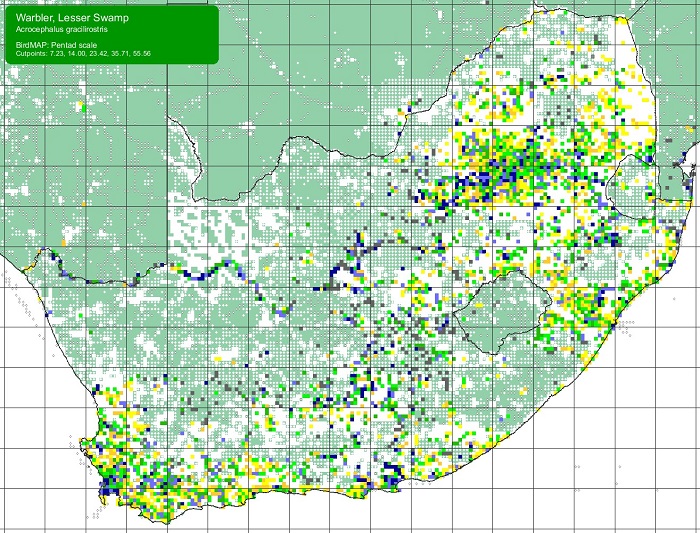
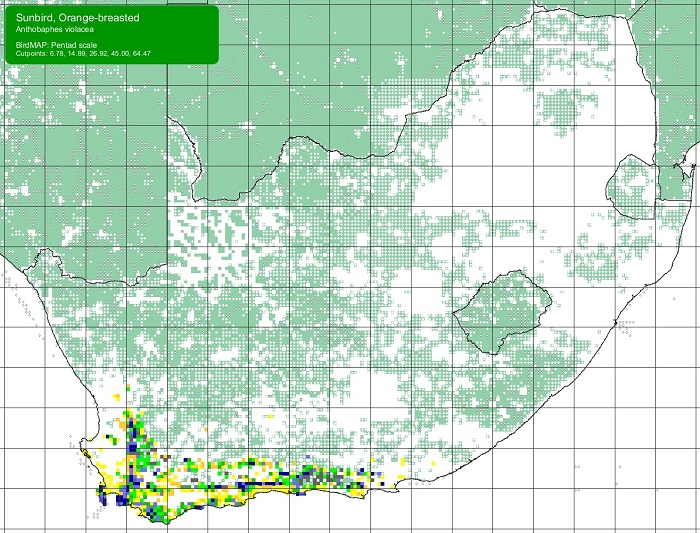
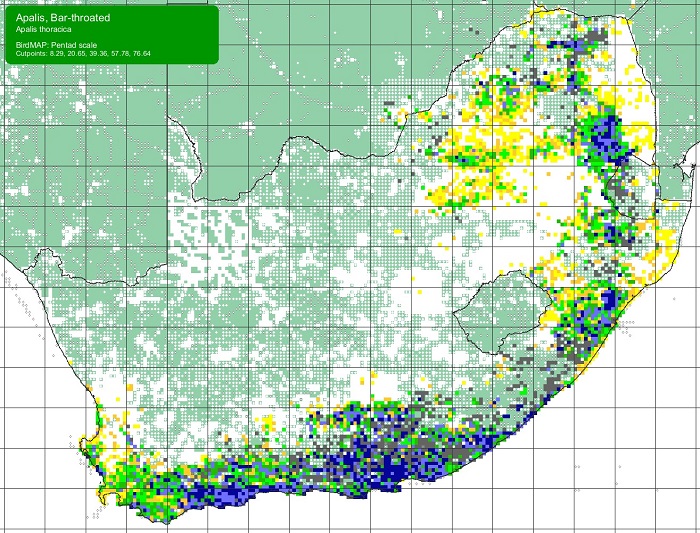
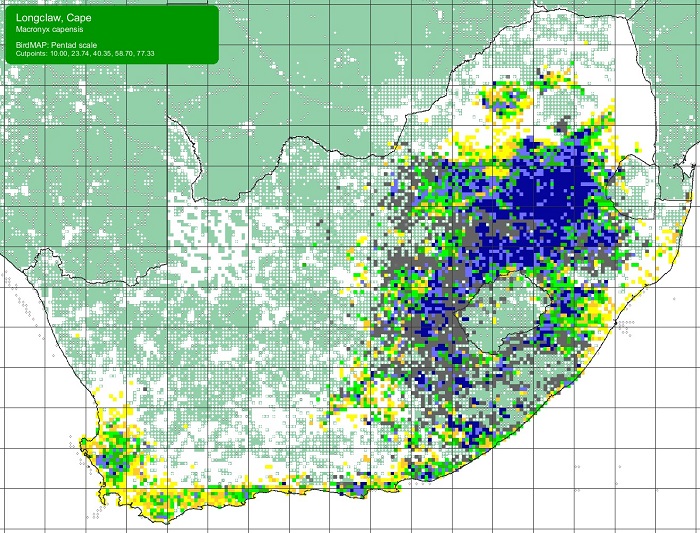
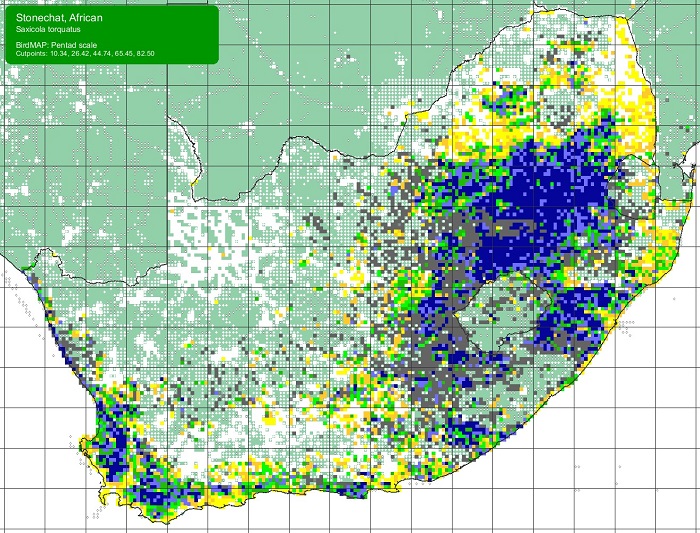
The Scaly-feathered Finch is a locally Common near-endemic to southern Africa. Beyond the region, its range extends only into south-western Angola. In southern Africa, it occurs throughout Namibia (except the coastal desert) and Botswana, where it has a stronghold in the central and southern Kalahari. It is also found in central and western Zimbabwe. Its South African range includes western Limpopo Province and north-western Mpumalanga, south and west to the Northern Cape, with a seemingly isolated population in the Eastern Cape interior and adjoining Western Cape.
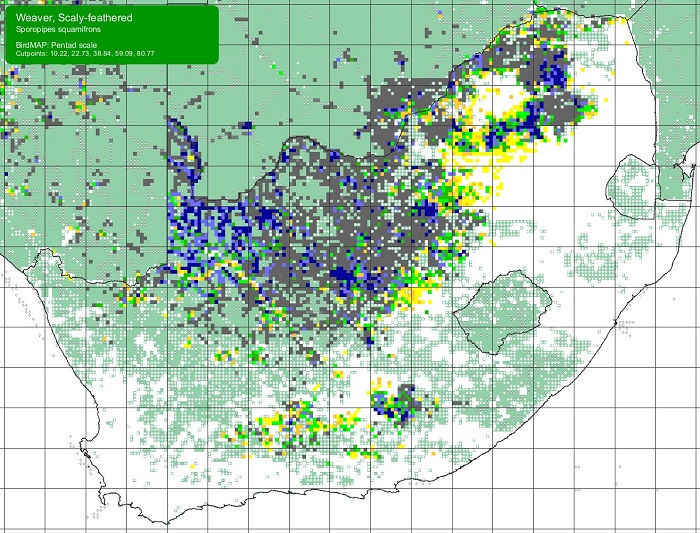
Details for map interpretation can be found here.
The Scaly Feathered Finch is not threatened, and its historical range is not known to differ from that of the present. It has benefited from thornbush encroachment due to overgrazing and is well represented in protected areas.

Near Carnarvon, Northern Cape
Photo by Ryan Tippett
Habitat
The Scaly-feathered Finch prefers dry, open woodland dominated by thorn trees (Vachellia & Senegalia) and favours areas with small trees and patches of grass. In more arid, open areas like the Nama Karoo and southern Kalahari, it inhabits shrubs and thickets along drainage lines. The Scaly-feathered Finch also frequents farmyards and gardens.

Mokala National Park, Northern Cape
Photo by Karis Daniel
Behaviour
The Scaly-feathered Finch is resident but becomes locally nomadic during dry periods. They occur in small groups of up to 20 birds throughout the year, but flock size often increases in winter and during droughts.

Near Pofadder, Northern Cape
Photo by Zenobia van Dyk
The Scaly-feathered Finch drinks water regularly when available. However, it is well adapted to arid environments and can survive without water for extended periods. It is an active species that forages on the ground. Groups hop about restlessly in their search for food. The diet consists mostly of grass seeds and is supplemented by termites.

Mokala National Park, Northern Cape
Photo by Terry Terblanche
At night, groups of Scaly-feathered Finches roost in thin-walled roosting nests; if disturbed, they burst through the roof of the nest before scattering. When available, they also roost in Sociable Weaver nests. The Scaly-feathered Finch is often tame and confiding.

Kgalagadi Transfrontier Park, Northern Cape
Photo by Andre Kok
The Scaly-feathered Finch is monogamous. They are possibly facultative cooperative breeders, where individuals (often young from previous years) help raise the young. However, this assistance is not essential for the species’ reproductive success. The Scaly-feathered Finch is known to breed opportunistically throughout the year in drier regions, but mostly during spring and summer in the more mesic eastern parts of its range. They are not colonial breeders, but nests may sometimes be clustered within a small area.

Danielskuil, Northern Cape
Photo by Aletta Liebenberg
The nest is a scruffy, hollow ball of pale dry grass stems and inflorescences, with a spout-like side entrance tunnel. The aromatic green stems of Pentzia spp shrubs are often incorporated into the structure and may serve as a fumigant to repel ectoparasites. It is then lined with fine grass flowers. The nest is situated up to 4m (usually lower) above the ground in the thin branches of a thorn bush or tree. 2 to 7 eggs are laid per clutch. The incubation period takes up to 12 days to complete. Thereafter, the nestling period takes. Scaly-feathered Finch broods suffer a fairly high predation rate. One study found that up to 30% of broods fail due to predation.

Near Boshof, Free State
Photo by Dawie de Swardt
Further Resources
This species text is adapted from the first Southern African Bird Atlas Project (SABAP1), 1997.
The use of photographs by Aletta Liebenberg, Andre Kok, Dawie de Swardt, Joanne Putter, Karis Daniel, Kyle Finn, Norman Barrett, Ryan Tippett, Sybrand Venter, Terry Terblanche, and Zenobia van Dyk is acknowledged.
Other names: Scaly-feathered Weaver (Alt. English); Baardmannetjie (Afrikaans); Sporopipe squameux (French); Baardmanwever (Dutch); Schnurrbärtchen (German); Tecelão-de-testa-malhada (Portuguese).
Recommended citation format: Tippett RM. 2026. Scaly-feathered Finch Sporopipes squamifrons. Biodiversity and Development Institute. Available online at https://thebdi.org/2026/03/18/scaly-feathered-finch-sporopipes-squamifrons/
List of bird species in this format is available here.

Mokala National Park, Northern Cape
Photo by Sybrand Venter