Cover photo by Megan Loftie Eaton.
Find the Rose’s MountainToadlet in the FBIS database (Freshwater Biodiversity Information System) here.
Family Bufonidae
Identification
Rose’s Mountain Toadlet is a small Bufonid with an elongated body that reaches a length of c.28 mm in males and 39 mm in females (Channing 2001). The dorsum is smooth and soft with scattered blister-like ridges and warts on the back and sides, while the posterior region of the ventrum has heavily granulated skin (Passmore and Carruthers 1995). The pupil is horizontally elliptical. Interestingly, this species lacks external or middle-ear structures (Grandison 1980) and apparently has no advertisement call. The parotid glands are distinct and have an inverted pear shape (when viewed from the rear). There is no webbing between the toes, and the hind limbs are relatively short and better adapted for walking and running than for jumping (Poynton 1964; Wager 1965).
The upper body surface is variably shaded in grey to brown, interspersed with darker spots and blotches. A light vertebral stripe or band and a pair of light dorsolateral bands are usually present. The parotid glands are generally orange to red and markings of the same colour may be present elsewhere on the upper body surface. The underside is white with variable grey shading and some darker markings may be present. These frogs often take on a uniformly blackish appearance when in dark surroundings.
Rose’s Mountain Toadlet most resembles C. magistratus and C. selenophos as these species also lack a tympanum and external or middle-ear structures. Two other Capensibufo species, namely, C. deceptus and C. tradouwi are also similar, but they posess a tympanum and produce advertisement calls (See ‘Similar Species’ below).

Silvermine, Western Cape
Photo by Luke Kemp
Habitat
Rose’s Mountain Toadlet is restricted to mountains where it occurs in undisturbed Mountain Fynbos. Although it is usually associated with the generally flatter topography on mountain tops, C. rosei has also been recorded from low-lying plateaus on the southern Cape Peninsula.
Breeding takes place in small, shallow, temporary pools of standing water that form during the winter rainy season. The pools can vary in size from c.20 × 20 × 2 cm deep to 250 × 150 × 6 cm deep. The vegetation surrounding breeding pools is usually dominated by restios. Pool substrates generally consist of a dark humic (peaty) or mossy layer on a sandy or rocky base and vary from light grey to almost black depending on the humic content. A breeding site can consist of one or more small pools in a seepage area of, for example, 100 m2. The species is not known to breed in pools of moving water associated with mountain streams.
Behaviour
Rose’s Mountain Toadlet has been seen in all four seasons of the year, but breeds only in late winter and spring. The commencement of breeding is dependent on rainfall and the formation of small, shallow pools of water. There are relatively few published records of C. rosei breeding activity. Spawn has been found in late July to early October.
An unusual characteristic of Capensibufo rosei is the absence of middle-ear elements (Grandison 1980), and it along with C. magistratus and C. selenophos appear to be the only southern African amphibians that lack a voice. Large, dense breeding aggregations and repeated use of the same site may facilitate meeting of the sexes. For example, at least 150–200 adults (and spawn) were observed in a partially divided breeding pool with overall dimensions of 250 × 150 cm and up to 6 cm deep (pers. obs.). (A similar observation was made by Power and Rose, who also described the eggs, tadpoles and metamorphosis of C. rosei (Rose 1962). During the breeding season, a bright pink, oval patch appears below the vent in both sexes (Power and Rose 1929), and a distinct swelling develops in the cloacal region in breeding males. The functions of these structures are not known.
These toads appear to be particular in their choice of breeding site. They have been found to use the same site on a few occasions during a breeding season, despite the presence of other similar small pools in the general vicinity (pers. obs.; southern Cape Peninsula).
The large, pigmented eggs are laid in a single transparent jelly tube that resembles a string of beads in that there is a distinct narrowing of the tube between the egg capsules (Power and Rose 1929). The clutch size is reported to be <100 eggs (Grandison 1980) but the typically dense breeding aggregations can lead to the deposition of thousands of eggs in a single pool. Adults in the breeding pool sometimes hide under the egg masses when disturbed.
The embryos take up to 12 days to leave the egg capsules. The tadpoles are dark in colour and attain a length of about 21 mm, of which more than half comprises the tail. They are benthic in habit, and have been observed feeding on egg capsules, decaying eggs and dead adults. Metamorphosis takes about six weeks, depending on temperature and the availability of food and water. The tiny toads are about 6.25 mm in length when they leave the water.
The predators and prey of this species have not been recorded.

Silvermine, Western Cape
Photo by Felicity Grundlingh
Status and Conservation
Status
C. rosei was previously listed as Restricted (Branch 1988) and is presently listed as Vulnerable (Harrison et al. 2001; this publication). It is legally protected by Nature Conservation Ordinance 19 of 1974, but is not listed by CITES.
Rose’s Mountain Toadlet has a restricted and fragmented distribution comprising about six subpopulations (sensu IUCN criteria). While most C. rosei habitat is situated in pristine mountain areas, habitat degradation has led to <20% habitat loss over the last 30 years. However, in some areas, the quality of C. rosei habitat is likely to have improved with systematic alien vegetation control programmes. For example, the previously degraded habitat of C. rosei in the Silvermine area on the Cape Peninsula (Boycott 1988e) now falls within Cape Peninsula National Park, and the pine plantations and dense stands of alien vegetation have been removed.
C. rosei is known to occur in the following protected areas: Cape Peninsula National Park (including the former Silvermine Nature Reserve and Cape of Good Hope Nature Reserve), Hottentots Holland, Kogelberg, Fernkloof, Maanschynkop, Vogelgat, Limietberg and Riviersonderend nature reserves.
Threats
In some places, one or more of the following factors have led to degradation or loss of habitat, or are a potential threat to populations of C. rosei. These include invasive alien vegetation, plantations, wildflower farming, alteration of drainage patterns, too frequent fires, building developments such as roads and dams, and urban development on the southern Cape Peninsula. The most pressing of these threats is probably the spread of invasive alien vegetation that eliminates fynbos vegetation and reduces water resources.
The tendency of C. rosei to form dense breeding aggregations at specific sites increases its vulnerability, since the destruction or degradation of a breeding site seriously threatens the continued existence of that local population.
Amphibian declines in some parts of the world have taken place in pristine, high-altitude areas, and this has led to speculation supported by some scientific evidence that global environmental factors are responsible (Beebee 1997). In particular, climatic and atmospheric changes brought about by widespread atmospheric pollution, increased ultraviolet radiation and global warming appear to be the prime causes. These factors may also lower amphibians’ resistance to disease. There is no evidence, at present, that global factors have caused declines in southern African amphibian populations, but such threats may be significant.
Recommended conservation actions
The distribution and conservation status of C. rosei is monitored by the Western Cape Nature Conservation Board (De Villiers 1997a) as part of a threatened species monitoring programme. This helps ensure that appropriate recommendations can be made to promote the conservation and wise management of this species and its habitat. Management of C. rosei habitat is included in the management plans of the respective protected areas and mainly concerns the control of invasive alien vegetation.
Research is underway to investigate the genetic relationships between the various geographically fragmented subpopulations of C. rosei. Life history studies of this species are also in progress. A captive breeding programme for C. rosei is not recommended.
Distribution
Rose’s Mountain Toadlet is endemic to the winter-rainfall region of the Western Cape Province. It has a limited and patchy distribution. Its populations are restricted to the Cape Peninsula and mountains southwest of the Breede River valley, which extends from the Tulbagh/Worcester valley south-eastward to Witsand on the south coast, and the Nuwekloof gap that separates the Bain’s Kloof Mountains from the mountains to the north (Grandison 1980). Recorded localities vary in altitude from 60 m on the southern Cape Peninsula to 1600 m, although >80% of localities are above 400 m. The areas in which this species occurs receive 600–>3000 mm of rain per annum.
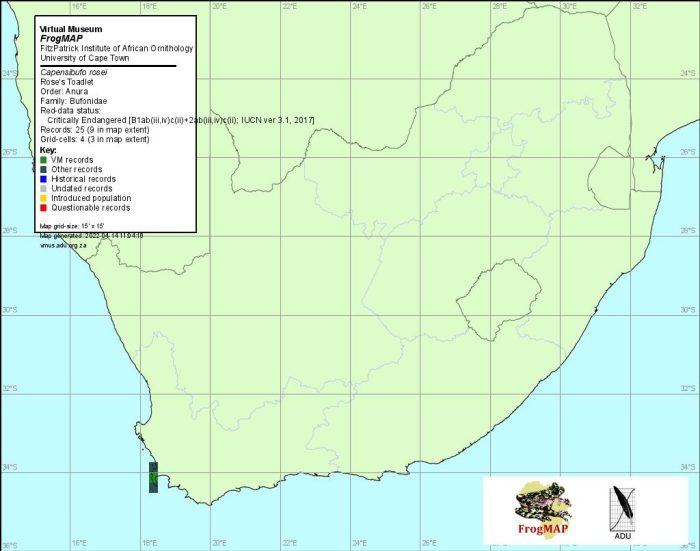
The Rose’s Mountain Toadlet was discovered on the Cape Peninsula (Hewitt 1926a), where it is known from the following localities: Table Mountain (3318CD), Muizenberg Mountain (type locality), Kalk Bay Mountain, Silvermine Valley, Chapman’s Peak and Kommetjie area (all in 3418AB), and Cape of Good Hope area (3418AD). However, the main part of its distribution range lies elsewhere, and includes the following localities: Bailey’s Peak in Limietberge Mountains (a.k.a. Bain’s Kloof Mountains) and Hawekwa Mountains (3319CA), Riviersonderend Mountains (3419BB) including Jonaskop (3319CD) and Kanonberg (3419BA), Klein River Mountains (3419AD), Jakkals River catchment area, Nuweberg Mountain near Viljoen’s Pass, Groenlandberge Mountains, Landdroskop in the Hottentots Holland Mountains (all in 3419AA), Elgin Forest Reserve, and Kogelberg State Forest (3418BB).
Capensibufo rosei has a geographically fragmented distribution pattern that, based on current knowledge, appears to comprise six populations: Table Mountain, southern Cape Peninsula, Hottentots Holland/Kogelberg mountain complex, Dutoitsberge/Limietberge mountain complex, Riviersonderend Mountains, and Klein River Mountains.
In summary, C. rosei has been found in a total of 10 quarter-degree grid cells, all of which were recorded for the first time before 1990. However, since 1996 the species has been recorded in only five of these cells: 3418AB, 3418AD, 3319CA, 3419AA and 3419AD. Most of the latter records were obtained in 2000 during a threatened frog monitoring programme (De Villiers 1997a).
This small, cryptic toad is difficult to find in its remote montane habitat. However, the distribution data are accurate and it is unlikely that the limits of the species’ range will be extended by further surveys.
Further Resources
The use of photographs by Felicity Grundlingh, Luke Kemp, and Megan Loftie Eaton is acknowledged.
Rose’s Mountain Toadlet Capensibufo rosei (Hewitt, 1926)
Other Common Names: Gestreepte Skurwepadda (Afrikaans)
Recommended citation format: de Villiers, AL; Tippett, RM. (2025). Rose’s Mountain Toadlet Capensibufo rosei. Biodiversity and Development Institute, Cape Town. Available online at https://thebdi.org/2021/12/20/roses-mountain-toadlet-capensibufo-rosei/
This species text has been updated and expanded from the text in the
2004 frog atlas: de Villiers, AL. (2004). Rose’s Mountain Toadlet Capensibufo rosei. In Minter LR et al 2004.
References:
Minter, LR; Burger, M; Harrison, JA; Braack, HH; Bishop, PJ; Kloepfer, D. (Editors). (2004). Atlas and Red Data Book of the Frogs of South Africa, Lesotho and Swaziland. Smithsonian Institution, Washington, and Avian Demography
Unit, Cape Town.
Carruthers, V; du Preez, L. (2017). Frogs of southern Africa: A Complete Guide. Struik Nature, Cape Town.
Channing, A. (2001) Amphibians of Central and Southern Africa. Protea Book House, Pretoria

