Cover image of Kori Bustard by Neels Putter – Mapungupwe National Park, Limpopo – BirdPix No. 257451
Bustards and Korhaans belong to the Family OTIDIDAE. They are robust, medium to very large ground birds. They have long necks and relatively large heads. The legs are long and the feet are equipped with 3 short, strong toes. The feet lack hind toes.
Identification
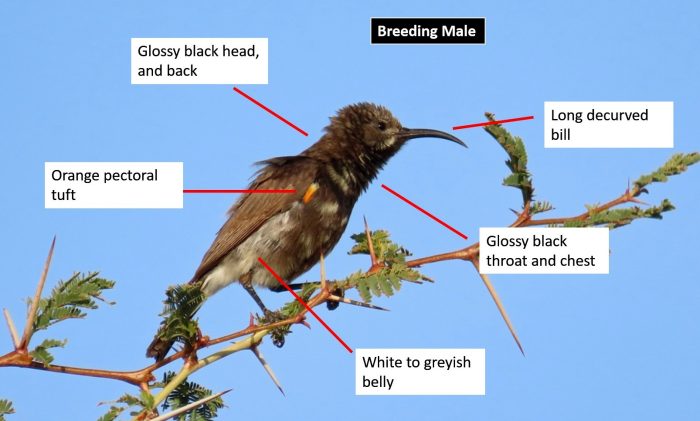
The Kori Bustard is a very large and conspicuous species. It is the largest of all bustards and is in fact the world’s heaviest flying bird. Male Kori Bustards are much larger than females. Males weigh 12.4kg on average, but can attain a weight of up to 19kg. The average weight of females is 5.7kg. The sexes differ very slightly in plumage coloration.

Kruger National Park, Limpopo
Photo by Ansie Dee Reis
In males, the crown is black, with an indistinct grey median stripe. A long crest on the hind-crown gives it a characteristic appearance. The face is grey, with a whitish supercilium that extends behind the eye. The throat is white and the feathers around the entire neck are finely barred grey and white. At the base of the hind neck there is a black collar that extends onto the sides of the breast. The rest of the underparts, from the breast to the vent are white.

Mashatu Game Reserve, Botswana
Photo by Derek Solomon
The upperparts are greyish-brown with fine dark brown flecks. The wing coverts form a variable black-and-white panel in the folded wing. Flight feathers are greyish-brown, with fine whitish bars. The tail is greyish-brown, with 2 creamy white bars towards the base. The bill is pale horn-coloured and the eyes are yellow. Leg colouration varies from yellow to dark cream.
The far smaller females closely resemble males, but show less black on the crown and have a narrower supercilium and a more finely barred neck. Juveniles are similar to adult females, but have a paler head, a shorter crest and a browner back.

Near Kotzesrus, Western Cape
Photo by Salome Willemse
The Kori Bustard can be confused with Denham’s Bustard (Neotis denhami) and the Ludwig’s Bustard (Neotis ludwigii). Both of these species are much smaller and have chestnut (not grey and white) hind necks, and they show striking white wing markings that are visible in flight. Additionally, both lack the dark crest and black collar.

Etosha National Park, Namibia
Photo by Roy Earle
Distribution
The Kori Bustard has a disjunct African distribution with two populations comprising two subspecies, one in southern Africa and the other in north-east Africa. It is widespread, although patchily distributed, in southern Africa.

It occurs mainly in the semi-arid regions in the western half of southern Africa but is largely absent from the extremely arid Namib Desert. The species penetrates to the east along the central plateau of Zimbabwe and the dry Limpopo River Valley into the eastern lowveld of South Africa, just reaching southern Mozambique. There is a partially isolated population south of the escarpment in the southern Nama Karoo between Prince Albert (Western Cape) and Makhanda (Grahamstown) (Eastern Cape). The largest populations occur in Namibia and Botswana.

Kruger National Park, Limpopo
Photo by Marius Meiring
The Kori Bustard is scarce to locally common in southern Africa and is currently listed as Vulnerable in South Africa. The Kori Bustard has decreased in range and abundance in several regions, including Zimbabwe and Eswatini (Swaziland) where it became extinct prior to 1960. In South Africa it has decreased markedly in Limpopo and Mpumalanga and is now absent from the central savannas of north-eastern South Africa. The species has also declined in the Eastern Cape province. The Kori Bustard is now common only in large protected areas. It is, however, still present in lower numbers in many smaller protected areas. The threats faced by this species are numerous and include habitat destruction, bush encroachment, high human densities, poisoning, deliberate hunting, collisions with overhead transmission lines and stray dogs.
Habitat
The Kori Bustard inhabits fairly dry, open habitats on flat terrain and with rainfall between 100 to 600 mm per year. Typical habitats include dry, open savanna, Nama Karoo dwarf shrublands and arid grasslands. They also frequent dry floodplains and the grassy edges of seasonal pans.

Near Vanwyksvlei, Northern Cape
Photo by Ryan Tippett
They are often found close to tree-lined watercourses, which provide cover when disturbed and shade during the heat of the day. The Kori Bustard is usually absent from areas of dense vegetation. They are frequently found on sandy soil, especially Kalahari sands, but also occur on stony ground. They are mainly encountered in natural vegetation, but sometimes aggregates in cleared areas, such as airstrips, fire-breaks, pastures and fields. They are occasionally attracted to burnt ground.
Behaviour
The Kori Bustard is most often found solitarily or in pairs when breeding. They are otherwise somewhat gregarious in groups of up to 40 or more. They spend most of their time walking slowly across open habitats. They walk or run quickly when disturbed but are usually reluctant to fly. The flight is heavy and laboured, yet powerful. Kori Bustards normally take flight after running a short distance but can take-off from a standing position if they need to. In hot weather they usually rest up in shade to avoid the mid-day heat.

Pilanesberg National Park, North West
Photo by Pieter Cronje
They drink regularly when water is available but are not dependant on water. Drinks with a sucking action, usually by lying down on the tarsal joint, allowing the head and neck to be almost level with the water.

Etosha National Park, Namibia
Photo by Aron White
Movements of the Kori Bustard are poorly understood and there is no evidence for regular migration. Any movements are more likely to be nomadic in response to the unpredictable rainfall of semi-arid areas.
Kori Bustards roost on bare ground or in very short grass where they remain crouched and immobile the entire night.

Kgalagadi Transfrontier Park, Northern Cape
Photo by John Tinkler
Forages by walking and pecking on the ground or in low bushes and trees. Kori Bustards are omnivorous and have a varied diet. They do not have a crop but possess well-developed caeca (secondary fermentation chambers) in the large intestine which indicates that plant material plays an important role in the diet. This allows them to consume green leaves, grass, flowers, roots, bulbs, seeds and berries. They also feed on wild melons and the gum of Vachellia (Acacia) trees, hence its Afrikaans name of ‘Gompou’.

Mapungubwe National Park, Mpumalanga
Photo by Neels Putter
Animal matter in the diet includes invertebrates like grasshoppers, locusts, beetles, termites, caterpillars, solifuges, scorpions and snails. Kori Bustards are especially fond of Dung Beetles and Armoured Ground Crickets. They also eat small vertebrates such as rodents, lizards, chameleons, snakes and birds’ eggs and nestlings. They also sometimes consume carrion. Kori Bustards are attracted to locust and caterpillar outbreaks and will visit recently burnt areas in the hopes of finding prey that may have perished in the flames. They can occasionally be seen foraging alongside Denham’s and Ludwig’s Bustards.
In some regions, notably the Savuti Depression in Botswana, Southern Carmine Bee-eaters (Merops nubicoides) are commonly seen to perch on the backs of Kori Bustards, in order to hawk flushed insects. However, this association is rarely reported elsewhere in southern Africa. There are also records of Fork-tailed Drongo (Dicrurus adsimilis) doing the same.

Savuti, Botswana. The bird on the bustard’s back is a Southern Carmine Bee-eater.
Photo by Pieter Cronje
Kori Bustards are polygynous, whereby males mate with multiple females through the breeding season. Males establish regularly used lek sites where they display competitively in order to entice females for mating. Leks are mostly situated in prominent positions such as on a rocky ridge. Several males may gather at these sites to display, but usually one male will achieve dominance over his rivals through complex posturing, forcing the others to move away. Evenly matched males may end up fighting which involves them standing chest-to-chest, their tails fanned and erect and their bills locked together. They then engage in ‘pushing’ one another for up to 30 minutes until a victor emerges.

Mapungubwe National Park, Limpopo
Photo by Neels Putter
Dominant males perform courtship displays mainly during the early morning and late afternoon. They strut about with their crests erected, tails fanned, winged drooped and necks inflated to 4 times the normal size. The neck feathers are long and loose to allow the neck to expand. They utter a booming, far carrying call when the neck is fully inflated. This sound is created by the air as it gets forcibly expelled from the inflated oesophagus. The inflated neck serves to resonate and amplify the booming call over a great distance. This display also serves a visual function and is conspicuous from some way off. Females choose the dominant males for mating and after copulation males take no further part in in the breeding process.

Mapungubwe National Park, Limpopo
Photo by Derek Solomon
No true nest is constructed. The nest is merely a shallow, unlined scrape on the ground, often in sandy soil. The nest site is sometimes reused in successive years. Usually 2 (sometimes just 1) eggs are laid per clutch. The eggs are pale olive-brown with darker olive-green and brown streaks and shading. Eggs are laid between July and April but peak laying times vary by region, and is likely linked to rainfall. The incubation period lasts for around 24 days, during which time the female will regularly turn the eggs with her bill. The highly precocial young are cared for by the female and may remain with her for up to 18 months if she does not breed again the following year.
Kori Bustards are long-lived and have been recorded to live up to 26 years.

Etosha National Park, Namibia
Photo by Johan Van Rooyen
Further Resources
This species text is adapted from the first Southern African Bird Atlas Project (SABAP1), 1997.
The use of photographs by Ansie Dee Reis, Aron, White, Derek Solomon, Johan Van Rooyen, John Tinkler, Marius Meiring, Neels Putter, Pieter Cronje, Roy Earle and Salome Willemse is acknowledged.
Virtual Museum (BirdPix > Search VM > By Scientific or Common Name).
Other common names: Gompou (Afrikaans); Iseme (Xhosa); Kgôri (Tswana); Koritrap (Dutch); Outarde kori (French); Riesentrappe (German); Abetarda-gigante (Portuguese).
Recommended citation format: Tippett RM 2024. Kori Bustard Ardeotis kori. Biodiversity and Development Institute. Available Online at https://thebdi.org/2024/06/12/kori-bustard-ardeotis-kori/
A list of bird species in this format is available here.

Kruger National Park, Mpumalanga
Photo by Neels Putter