View the above photo record (by Trevor Hardaker) in FrogMAP here.
Find the Common Platanna in the FBIS database (Freshwater Biodiversity Information System) here.
Family Pipidae
COMMON PLATANNA – Xenopus laevis
(Daudin, 1802)
Identification

Near Lilongwe, Malawi
Photo by Gary Brown
Habitat
This species inhabits all of the biomes in the atlas region. Prior to the advent of modern agriculture, X. laevis probably occurred in low densities in natural water bodies, such as streams, rivers and their pools. Nowadays, however, the species is also found in a variety of man-made water bodies such as farm dams, ponds, sewage purification works and fish farms. Eutrophic waters seem to produce the highest densities.
There are some studies of native populations (Schoonbee et al. 1992), although the best descriptions of habitat are in respect of feral populations (Tinsley and McCoid 1996; Measey 2001). Breeding and non-breeding habitats appear to be the same, although there are no records of breeding in flowing water.
Behaviour
After heavy rains, X. laevis sometimes leave water bodies en masse, and single individuals are also encountered on the surface in damp weather. These appearances may be associated with movement to and from breeding sites (Du Plessis 1966). Breeding begins at the onset of the rains, thus at different times in the summer and winter rainfall areas (Berk 1938; Kalk 1960). There is a prolonged breeding period throughout the rainy season, and both females and males are able to breed more than once per season (Hey 1949).
Many laboratory studies have documented calling in X. laevis, describing the advertisement call of the male and acceptance or rejection by females. However, such studies have relied on hormonal induction and may not represent natural behaviours (Picker 1980; Kelley 1996; Tobias et al. 1998). Field studies have suggested that males call around the edges of territories, although this may be density dependent (A. Elepfandt pers. comm.). Spawning takes place during the night when couples, in inguinal amplexus, swim around the pond depositing single eggs on any hard substrate (McCoid 1985).
Larvae hatch within two to three days and, after finishing the yolk supply, begin to feed on algae suspended in the water column. Tadpoles display coordinated schooling behaviour, and maintain their position in the water column by means of a characteristic undulating motion of the tail (Wassersug 1996). Time to metamorphosis varies with temperature and the abundance of food. In optimal conditions, metamorphosis is possible within two months (Tinsley et al. 1996).
Adults may move from water bodies after breeding, reducing the incidence of cannibalism (Hey 1949; McCoid and Fritts 1980; Measey 1998b). Adults are generalist predators and scavengers, and can hold food items in their toothed mouths while breaking it apart with their claws using an overhead kick (Avila and Frye 1978). These behaviours can be detected by other adults in the vicinity and sometimes lead to a feeding frenzy (Frye and Avila 1979). Most food items for post-metamorphic X. laevis are benthic macro-invertebrates, such as chironomid larvae. However, a wide variety of food sources are used from all microhabitats in water bodies, including carrion and terrestrial food items (Measey 1998a, b). Even the largest animals take very small prey items, such as zooplankton and ostracods.
Toward the peak of the dry season, X. laevis will either move from drying water bodies or burrow into the wet mud to aestivate. Longevity is unknown for native animals, but in feral populations and in captivity, individuals are known to have lived for more than 15 years (Measey and Tinsley 1998).
X. laevis plays an important role in the ecology of southern African wetlands because it is widespread and abundant, and it is a voracious predator as well as an important prey item for several mammalian, avian and reptilian predators.
Status and Conservation
X. laevis does not seem to be threatened in any part of its range. Montane populations may be genetically distinct (Measey and Channing 2003; Grohovaz et al. 1996) and may warrant management attention. Hybridisation occurs at the northern and southern ends of its range, with X. muelleri and X. gilli, respectively.
X. laevis seems to present a problem to other species because of its invasive tendencies. This is exacerbated by the fact that thousands of these frogs have been exported from South Africa since the 1930s, and still are, because of the popularity of the species as a laboratory animal. There is concern that this trade may also be contributing to the global spread of chytridiomycosis (Weldon 2002).
Distribution
This species is widely distributed in sub-Saharan Africa. Six subspecies are recognised, but parasitological and molecular data indicate that X. laevis laevis is the most divergent of these taxa and should be raised to the species level (Kobel et al. 1996; Kobel et al. 1998; Jackson and Tinsley 1997; Measey and Channing 2003). X. l. laevis occurs throughout southern Africa south of the Zambezi River, and is the only subspecies currently known to occur within the atlas region. However, winter and summer rainfall areas hold genetically distinct groups that may represent different subspecies of X. laevis (Grohovaz et al. 1996; Measey and Channing 2003).
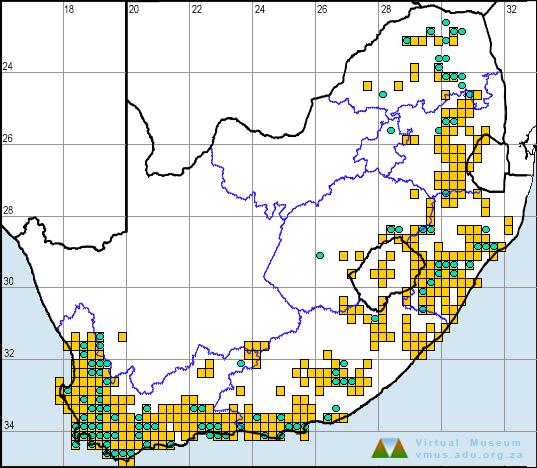
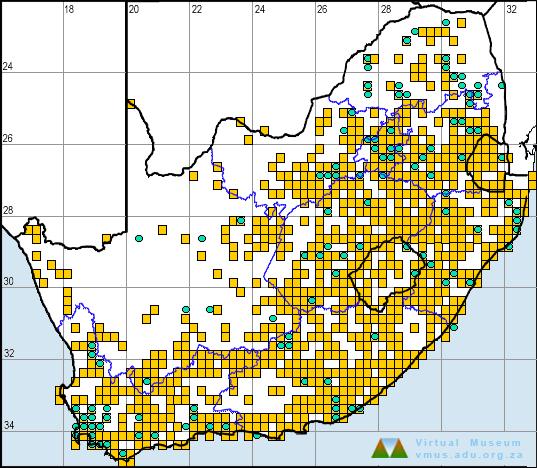
In the atlas region, X. laevis is a common and widespread species, occurring from sea level to nearly 3000 m in Lesotho. In the west, it is apparently absent in areas of extreme aridity, including much of the Kalahari and Bushmanland in Northern Cape Province, although this may be due to inadequate sampling. Its distribution extends eastward as far as the Great Escarpment, where it comes into contact with X. muelleri in the low-lying parts of Limpopo and Mpumalanga provinces (see X. muelleri account).
X. laevis is a highly invasive species, as is evidenced by the feral populations that have become established in many parts of the world. Its present distribution in the atlas region may not represent its ‘natural range’ as this frog is commonly used as live bait by fishermen and may have been inadvertently translocated to areas from which it was previously absent. The proliferation of farm dams and reservoirs over a few hundred years is another factor which may have enabled this species to expand its range.
The atlas data are reliable, but many of the gaps in distribution do not necessarily reflect absence.
Further Resources
Virtual Museum (FrogMAP > Search VM > By Scientific or Common Name)
More common names: Gewone Platanna (Afrikaans)
Recommended citation format for this species text:
Measey GJ, Tippett RM. Common Platanna Xenopus laevis. BDI, Cape Town.
Available online at http://thebdi.org/2021/11/08/common-platanna-xenopus-laevis/
Recommended citation format:
This species text has been updated and expanded from the text in the
2004 frog atlas. The reference to the text and the book are as follows:
Measey GJ 2004 Xenopus laevis Common Platanna. In Minter LR
et al 2004.
Minter LR, Burger M, Harrison JA, Braack HH, Bishop PJ, Kloepfer D (eds)
2004. Atlas and Red Data Book of the Frogs of South Africa, Lesotho and
Swaziland. Smithsonian Institution, Washington, and Avian Demography
Unit, Cape Town.