View the above photo record (by Faansie Peacock) in FrogMAP here.
Find the Knysna Leaf-folding Frog in the FBIS database (Freshwater Biodiversity Information System) here.
Family Hyperoliidae
KNYSNA LEAF-FOLDING FROG – Afrixalus knysnae
(Loveridge, 1954)
Identification
The presence of a vertical pupil and minute asperities (spines), distributed uniformly over the dorsal, and posterior ventral skin of the body, and the dorsal skin of the limbs, distinguish A. knysnae from the morphologically similar, and sympatric, Hyperolius marmoratus and H. horstockii. The asperities are more conspicuous in males than in females. Body length is 22–25.2 mm (Pickersgill 1996).
A. knysnae does not occur in sympatry with any other Afrixalus species. A. spinifrons has a shorter body (16.4–22.6 mm; Pickersgill 1996). In A. knysnae the snout is not noticeably swollen or bulbous, nor is there a concentration of tubercular asperities in this region, as is the case in most male A. s. spinifrons (but not A. s. intermedius). The gular disc is smaller and transversely oval to round in A. knysnae whereas, in both subspecies of A. spinifrons, the disc covers most of the gular surface (Pickersgill 1996).
The following description of the advertisement call is based on calls recorded at Covie on 17 September 2002 at 15–20°C (H. Braack and M. Burger unpubl. data). These calls consisted of two parts: a short “zip” and a longer “trill”, as in A. delicatus and A. spinifrons. The duration of 13 trills from three individuals was 0.16–2.64 s; the pulse rate was 24–29 pulses/s; and the emphasized frequency was 2.9–3.6 kHz. Three zip calls from one individual had a duration of 0.06–0.09 s; a pulse rate of 209–230 pulses/s; and an emphasized frequency of 3.2–3.3 kHz.
Larger samples of advertisement calls from single populations, recorded during moderate- to high-intensity choruses, and adjusted to a common air temperature and body mass, are needed to differentiate more clearly between the calls of A. knysnae, A. delicatus and A. spinifrons.

Photo by Rian Stander
Habitat
A. knysnae inhabits a coastal mosaic of Mountain Fynbos and Afromontane Forest in the Outeniqua District centre of endemism of the Coastal Mountain biogeographical province (Branch and Hanekom 1987). FitzSimons (1946) recorded specimens in glades, clearings and roadside pools at Diepwalle (= Deepwalls; 3323CA), while Pickersgill (2000) collected juveniles from “arum blooms on boggy ground near an irrigation dam at Barrington” (3322DD). A few of the known localities are situated in pristine habitat but the remainder have been subjected to varying degrees of habitat alteration and usually contain a high proportion of alien plant species.
Behaviour
The biology of A. knysnae is poorly known. Breeding activity has been recorded October–December at Covie, Groenvlei and Diepwalle, and in late February in the De Vasselot Nature Reserve west of Covie. Juveniles were collected in early October at Covie, hence breeding activity probably starts in September at this locality (FitzSimons 1946; Branch and Hanekom 1987; H. Braack unpubl. data). A female, collected at Diepwalle in early December, contained well-developed ovarian eggs (Pickersgill 1996, 2000).
During the breeding season, males were seen calling from fallen branches and emergent vegetation just above the surface of small pools along the roadside and in forest clearings (FitzSimons 1946), while juveniles were collected from restio stems along the edges of small pans in fynbos (Branch and Hanekom 1987). During the day, adults were found on vegetation surrounding these pans. Tadpoles collected at Grootkop, Knysna, were similar in morphology to those of A. s. spinifrons from Port St Johns (Inger 1989).

Photo by Armin Du Preez
Status and Conservation
Status
A. knysnae was classified as Data Deficient (Harrison et al. 2001), but additional data and reassessment have placed it in the Endangered category on the basis of an extent of occurrence <5000 km2, an area of occupancy <500 km2, a severely fragmented range, and a perceived continuing decline in the area of occupancy, numbers of individuals and locations, as well as a decrease in the area and quality of its habitat (this publication).
Threats
The long-term survival of this species is threatened by habitat loss and alteration as a result of extensive coastal urban and recreational development, forestry and other agriculture. An important factor linked to the development of the area is rapid growth of the tourism industry. Development has resulted in the draining, impoundment and eutrophication of wetlands near residential areas and agricultural lands, and encroachment of invasive alien vegetation. Impounded wetlands are often stocked with alien fish species and this may also have resulted in local extinction of A. knysnae populations.
The breeding site at Covie, a small seasonal pan, now lies within a pasture and most of the emergent vegetation has been destroyed by cattle grazing and trampling. Groenvlei is gradually becoming more brackish (R. Hiseman pers. comm.; monitored by the Department of Water Affairs and Forestry). These threats are continuing and are likely to lead to further population declines, possibly by as much as 20% or more in the next 30 years.
Recommended conservation actions
A. knysnae is currently protected within the Tsitsikamma National Park (Carruthers and Robinson 1977; Branch and Hanekom 1987) and the Goukamma Nature Reserve (including Groenvlei). Some sections of the Diepwalle forestry area may offer sanctuary, but the species was not recorded from these historical localities during atlas surveys in 2000 and 2001. The collection and trade of specimens are unlikely to be important threats; legislation should rather endeavour to protect the species’ habitat.
The plight of A. knysnae should be highlighted so that future environmental impact assessments (EIAs) within its range can attempt to prevent further loss of habitat. Private landowners should be encouraged to participate in its protection.
Conservation authorities should carry out surveys to find and quantify breeding populations of A. knysnae. All known localities should be assessed and afforded protection accordingly. It is strongly recommended that long-term monitoring be conducted at selected sites to track population fluctuations and identify limiting factors.
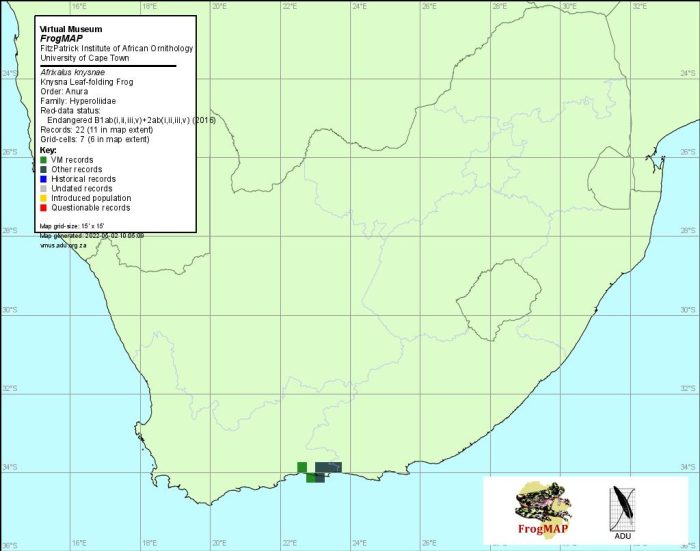
Distribution
Not all workers are in agreement about the extent of occurrence of A. knysnae. The maps of Poynton (1964), Passmore and Carruthers (1995) and Channing (2001), for example, include the central to northern Eastern Cape coastal belt as part of this species’ range. A. knysnae is difficult to distinguish from A. spinifrons, and further surveys and field studies are required to confirm the distributions of these two taxa. For the purposes of this atlas we have followed the assessment of Pickersgill (1996, 2000). As such, A. knysnae is endemic to the Western Cape Province. It occurs from Groenvlei (3422BB) in the west to Covie (3323DC) in the east, and is confined to the coastal region by the Outeniqua and Tsitsikamma mountains. A. knysnae is separated from the nearest A. s. spinifrons populations at Kei Road (3227DA) and Cintsa Bay (3228CC) by about 400 km, including at least 250 km of unsuitable habitat.
The atlas data are reliable but more intensive surveys are necessary to comprehensively map the distribution of this species.
Further Resources
Virtual Museum (FrogMAP > Search VM > By Scientific or Common Name)
More common names: Knysna-blaarvoupadda (Afrikaans)
Recommended citation format for this species text:
Braack HH, Minter LR, Tippett RM. Knysna Leaf-folding Frog Afrixalus knysnae. BDI, Cape Town.
Available online at http://thebdi.org/2021/11/16/knysna-leaf-folding-frog-afrixalus-knysnae/
Recommended citation format:
This species text has been updated and expanded from the text in the
2004 frog atlas. The reference to the text and the book are as follows:
Braack HH, Minter LR 2004 Afrixalus knysnae Knysna Leaf-folding Frog. In Minter LR
et al 2004.
Minter LR, Burger M, Harrison JA, Braack HH, Bishop PJ, Kloepfer D (eds)
2004. Atlas and Red Data Book of the Frogs of South Africa, Lesotho and
Swaziland. Smithsonian Institution, Washington, and Avian Demography
Unit, Cape Town.






























