Cover image of Orange-breasted Sunbird by Cobus Elstadt – Cape Peninsula, Western Cape
Sunbirds belong to the Family NECTARINIIDAE. This is a group of small passerine birds They are small, slender passerines from the Old World, usually with downward-curved bills. Many are brightly coloured, often with iridescent feathers, particularly in the males. A number of species also have elongated tail feathers. Sunbirds are found mainly in the warmer southern regions of the Old World. Species diversity is highest in equatorial regions. The family contains around 151 species from 16 genera.
Identification
The Orange-breasted Sunbird is a distinctive, colourful, and attractive species. It is the only member of its genus. The sexes differ markedly in plumage coloration.

Near Somerset West, Western Cape
Photo by Sybrand Venter
Adult males are easy to identify. The head, neck, upper mantle, and upper scapulars are metallic green. The rest of the upper parts are olive green. The brown tail is long and graduated with projecting central rectrices. They have bright yellow pectoral tufts, but these are infrequently visible in the field. There is a narrow violet band on the upper breast, while the remainder of the breast is rich orange, fading to yellow on the belly. The undertail coverts are also orange. The bill, legs and feet are black and the eyes are dark brown.

Table Mountain National Park, Western Cape
Photo by Jon Blanco
Adult females are drab in comparison. They are olive green, tinged with grey and brown overall. They sometimes have an orangeish wash on the belly. The tail is dark brown, with yellowish edges to the feathers. Juveniles are similar to adult females before male plumage begins to show.

Table Mountain National Park, Western Cape
Photo by Itxaso Quintana
Confusion with other species is only likely with females. The female Southern Double-collared Sunbird and Greater Double-collared Sunbird have less yellow breasts. The female Malachite Sunbird is far larger and has a longer bill.
Status and Distribution
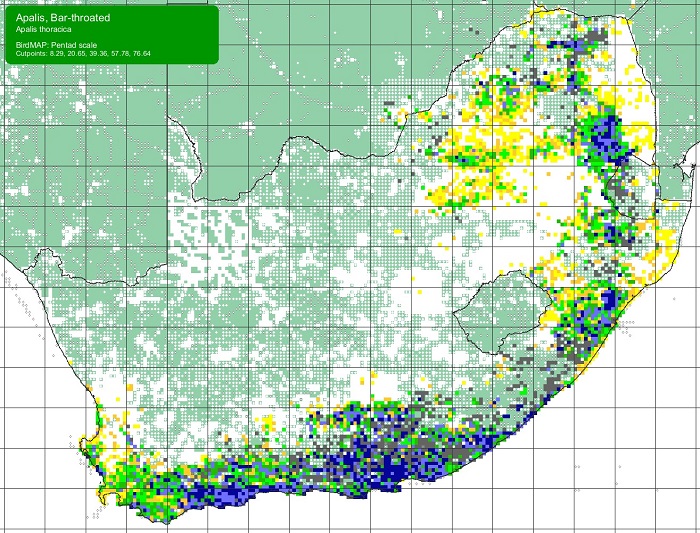
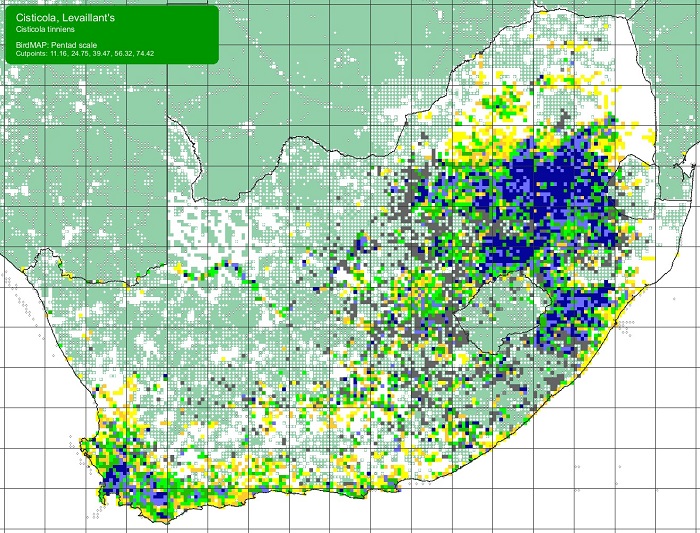
The Orange-breasted Sunbird is endemic to South Africa and to the Fynbos Biome. Its distribution extends from Vanrhynsdorp to Gqeberha (Port Elizabeth), but it is absent or scarce away from mountains along the west coast.
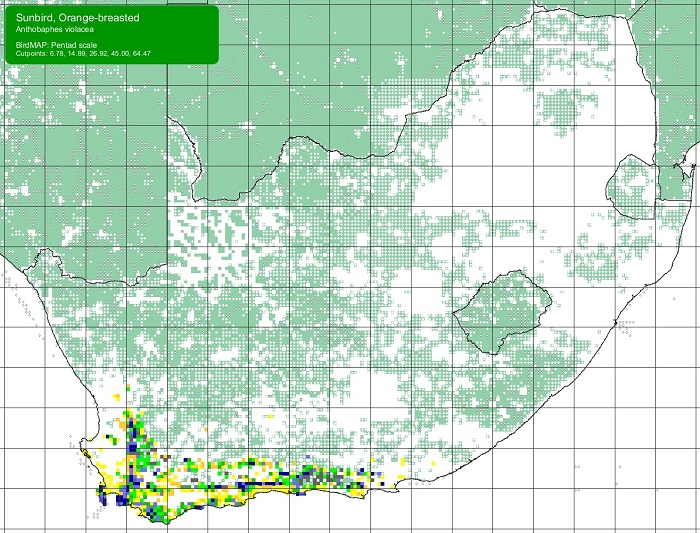
Details for map interpretation can be found here.
The Orange-breasted Sunbird is common and is not threatened. There is no evidence of any recent large-scale changes in its distribution. However, the Orange-breasted Sunbird is adversely affected by infestations of alien woody plants, too frequent burning, and, in the coastal lowlands, urban and agricultural development.

Mossel Bay, Western Cape
Photo by Joanne Putter
Habitat
The Orange-breasted Sunbird is endemic to the Fynbos Biome. It is restricted to fynbos shrublands and occurs from sea-level to high mountain peaks. The Orange-breasted Sunbird is found wherever plants of the genera Protea, Mimetes, Leucospermum, Erica, and many other plants with tubular flowers are in bloom. The Orange-breasted Sunbird is sometimes also found in gardens adjoining fynbos, but it is generally reluctant to leave fynbos. Nevertheless, the Orange-breasted Sunbird does occasionally venture beyond its traditional habitat and has been recorded in the Karoo, many kilometres from the nearest fynbos.

Swartberg Pass, Western Cape
Photo by Ryan Tippett
Behaviour
In common with other nectivores, the Orange-breasted Sunbird tracks the flowering of food plants. Its movements are poorly known, however, in large part due to the mountainous and remote nature of its habitat. Observation records indicate that birds move away from lowlands and return to higher altitudes in summer. Orange-breasted Sunbirds are sometimes forced to move into other areas in response to fire, including areas not normally frequented.

Kirstenbosch Botanical Gardens, Western Cape
Photo by Sybrand Venter
The Orange-breasted Sunbird is encountered singly or in pairs during the breeding season, but occurs in loose flocks of up to 100 birds at rich food sources in the non-breeding season. Drinks water and bathes frequently, along quiet reaches of streams or in dew that has accumulated on leaves.

Kogelberg Biosphere Reserve, Western Cape
Photo by Sharon Stanton
The Orange-breasted Sunbird consumes nectar and invertebrates, although nectar is its preferred diet. The Orange-breasted Sunbird is closely associated with tubular-flowered Erica (Heath) species, with which it appears to have co-evolved. The Orange-breasted Sunbird is the primary pollinator of Ericas. It also feeds on the nectar of a wide range of other plant genera, including Protea, Leucospermum, Leonotis, Mimetes, Watsonia, Lobostemon, Tecoma, and Aloe, etc. They also feed on the nectar of many alien plant species.

Kirstenbosch Botanical Gardens, Western Cape
Photo by Felicity Ellmore
The Orange-breasted Sunbird is often seen to forage on the ground for arthropods. It also carefully searches for invertebrates among leaves and branches, and frequently hawks insects in flight. Invertebrates eaten include spiders and a variety of insects like flies, wasps, beetles, grasshoppers, ants, termites, and cockroaches.
The Orange-breasted Sunbird is aggressive towards conspecifics and other birds around food sources. It is, however, often displaced from Protea flowers by male Cape Sugarbirds.

Table Mountain National Park, Western Cape
Photo by Itxaso Quintana
The Orange-breasted Sunbird breeds from February to November, with a peak between May and August. Differences in the timing of nesting across relatively short distances presumably reflect fine-scale variation in rainfall and/or flowering phenology of food plants.
The nest is a rounded oval with a side-top entrance. It is composed of rootlets, fine, leafy twigs, and grass, all of which are bound together with spider web. The nest is lined with brown protea fluff and other forms of plant down. It is usually situated in a proteaceous shrub, less than 1m above the ground. Nests may occasionally be found up to 10m above the ground in a tree. The nest is built entirely by the female.

Table Mountain National Park, Western Cape
Photo by Itxaso Quintana
1 to 2 eggs are laid per clutch. The eggs vary from whitish to grey-green and are blotched and mottled with shades of brown and grey. The incubation period takes 14 or 15 days and is done by the female only. The duration of the nestling period ranges from 15 to 22 days, during which time the young are fed by both parents. Fledglings return to the nest for up to 15 days after their initial departure.
Broods of the Orange-breasted Sunbird are sometimes parasitised by the Klaas’s Cuckoo Chrysococcyx klaas.

Near Stilbaai, Western Cape
Photo by Derick Oosthuizen
Further Resources
This species text is adapted from the first Southern African Bird Atlas Project (SABAP1), 1997.
The use of photographs by Cobus Elstadt, Derick Oosthuizen, Felicity Ellmore, Itxaso Quintana, Joanne Putter, Jon Blanco, Rick Nuttall, Sharon Stanton, and Sybrand Venter is acknowledged. Additional photographs by Ryan Tippett.
Other common names: Nectarinia violacea (alt. Scientific Name); Oranjeborssuikerbekkie (Afrikaans); Souimanga orangé (French); Oranjeborst-honingzuiger (Dutch); Goldbrust-Nektarvogel (German); Beija-flor-de-peito-laranja (Portuguese).
Recommended citation format: Tippett RM. 2026. Orange-breasted Sunbird Anthobaphes violacea. Biodiversity and Development Institute. Available online at https://thebdi.org/2026/02/20/orange-breasted-sunbird-anthobaphes-violacea/
List of bird species in this format is available here.

Addo Elephant National Park, Eastern Cape
Photo by Rick Nuttall