Cover photo by Desire Darling.
Find the Flapper Hooktail in the FBIS database (Freshwater Biodiversity Information System) here.
Family Gomphidae
Identification

Ndumo Game Reserve, KwaZulu-Natal
Photo by Ryan Tippett
Medium-large size
Length reaches 54mm;
The male Flapper Hooktail Paragomphus sabicus is readily identified by the combination of its greenish-yellow and black colouration, large black-brown foliations, and the elongate, hooked upper claspers. Females are similar to the males but lack the hooked claspers and carry smaller foliations on the abdomen.
The Flapper Hooktail most resembles the Rock Hooktail Paragomphus cognatus, but that species is noticeably smaller and the hooked upper claspers are black-tipped and outwardly splayed. The Rock Hooktail also bears noticeably smaller and paler tail foliations.
Click here for more details on identification of the Flapper Hooktail.

Ndumo Game Reserve, KwaZulu-Natal
Photo by Ryan Tippett
Habitat
The Flapper Hooktail is a species of large, flowing savanna rivers with well-wooded banks and usually an abundance of reeds. Individuals are most likely to be found away from the water in the surrounding woodlands above the riverbank.

Photo by Ryan Tippett
Behaviour
The Flapper Hooktail is seldom seen at the water’s edge. Both sexes of the Flapper Hooktail are most often found perched among trees and bushes above the riverbank. This species hunts from a perch, making swift flights to intercept prey before returning to settle once again. It can be fairly tame and confiding.
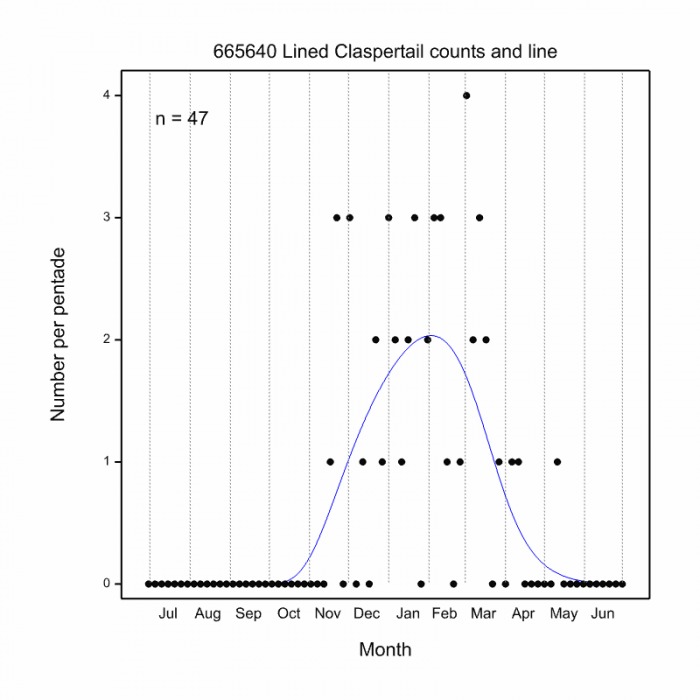
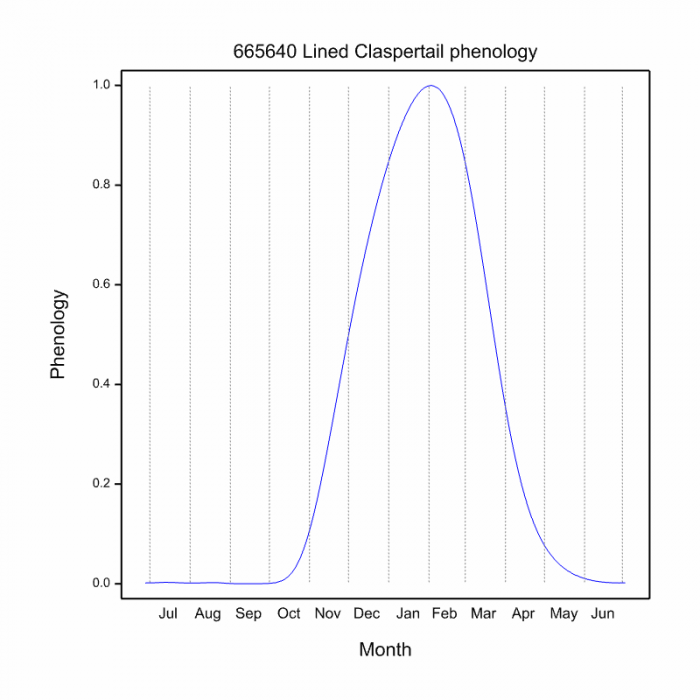
The Flapper Hooktail flies mostly from November to March (See Phenology below).

Ndumo Game Reserve, KwaZulu-Natal
Photo by Ryan Tippett
Status and Conservation
The Flapper Hooktail is uncommon to rare and localised in South Africa. Overall, it is listed as of Least Concern in the IUCN Red List of Threatened Species. In South Africa, the Flapper Hooktail is only known from undisturbed habitats.

Ndumo Game Reserve, KwaZulu-Natal
Photo by Ryan Tippett
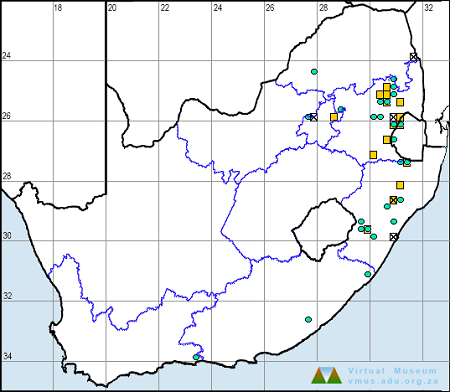
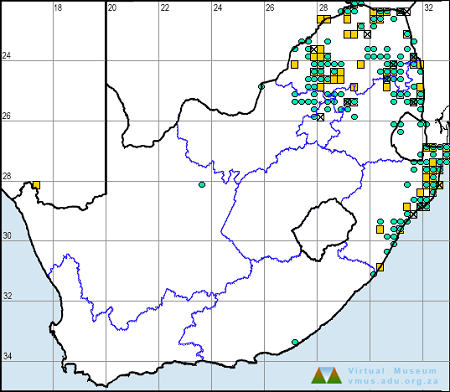
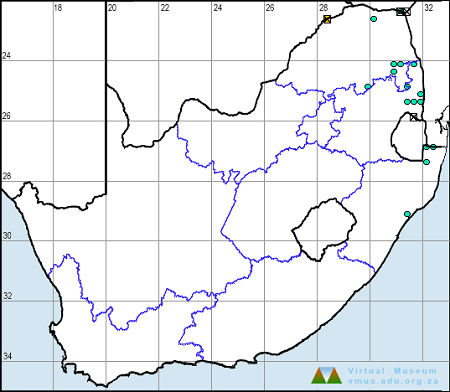
Distribution
The Flapper Hooktail is sparsely and erratically distributed across its range, occurring mainly in Southern and East Africa. It ranges from north-eastern South Africa up through parts of Zimbabwe, Northern Botswana, and Namibia to Zambia, Malawi, Tanzania, and Kenya. It has also been recorded once from the Nile River in Sudan.
In South Africa, the Flapper Hooktail is restricted to hot, low-lying areas in the Limpopo River valley, the Lowveld, and north-eastern KwaZulu-Natal.
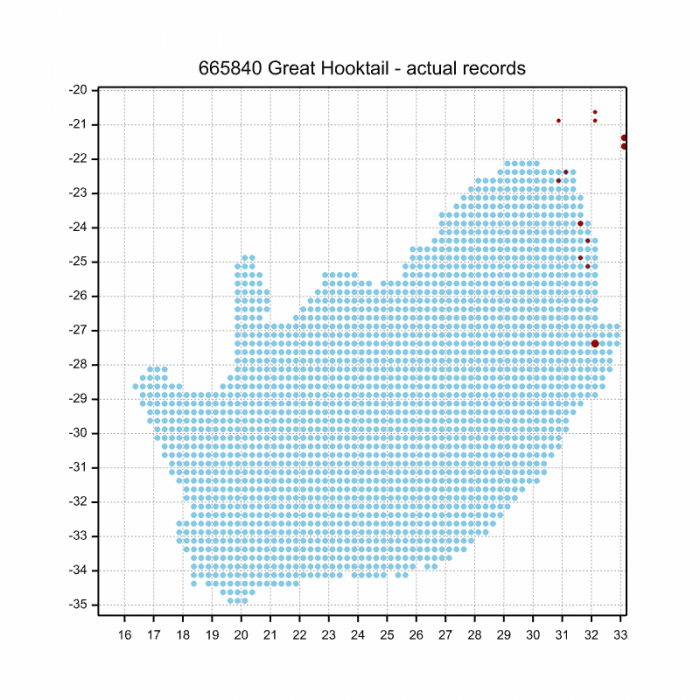
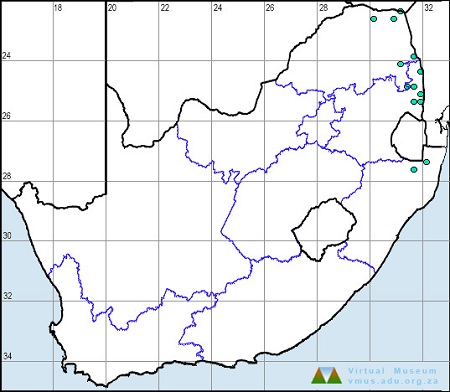
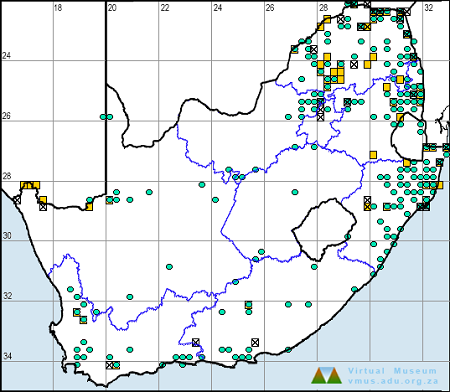
Below is a map showing the distribution of records for Flapper Hooktail in the OdonataMAP database as at February 2020.

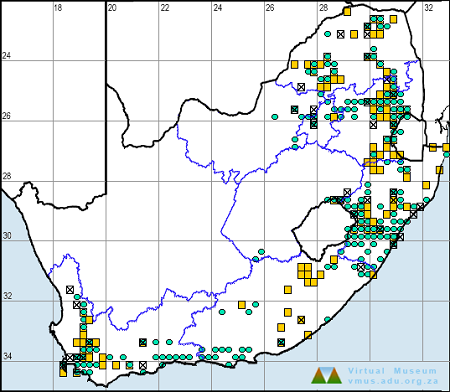
Below is a map showing the distribution of records for Flapper Hooktail in the OdonataMAP database as at December 2024.
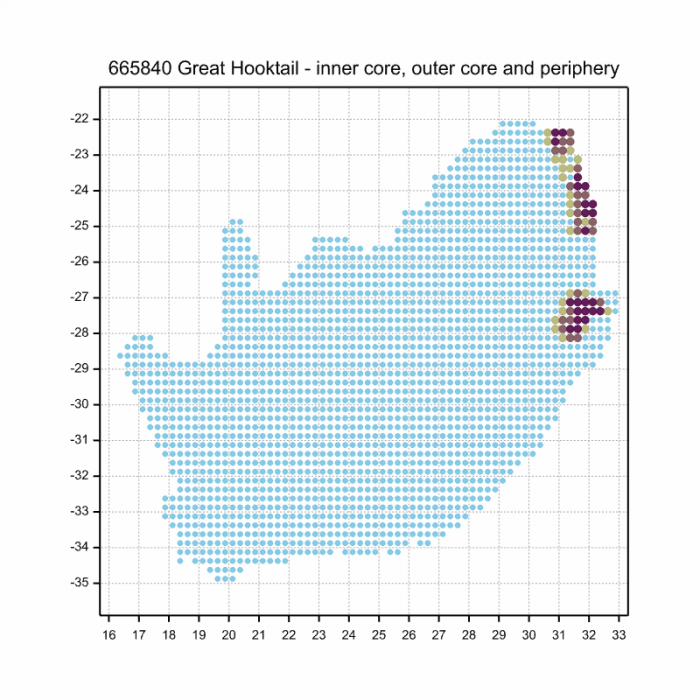
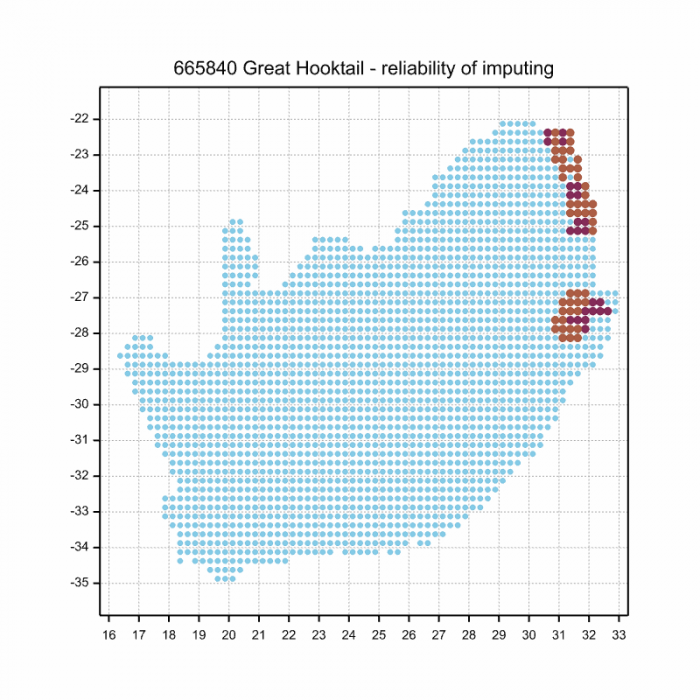
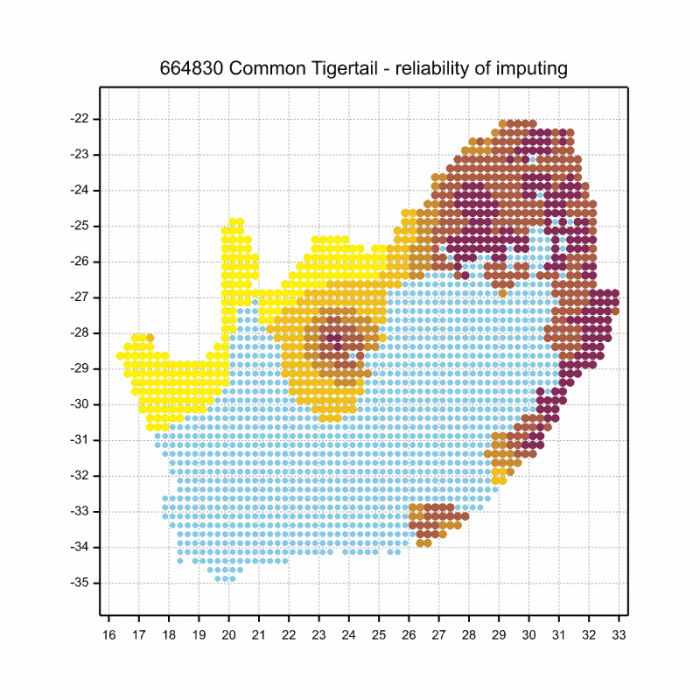
The next map below is an imputed map, produced by an interpolation algorithm, which attempts to generate a full distribution map from the partial information in the map above. This map will be improved by the submission of records to the OdonataMAP section of the Virtual Museum.

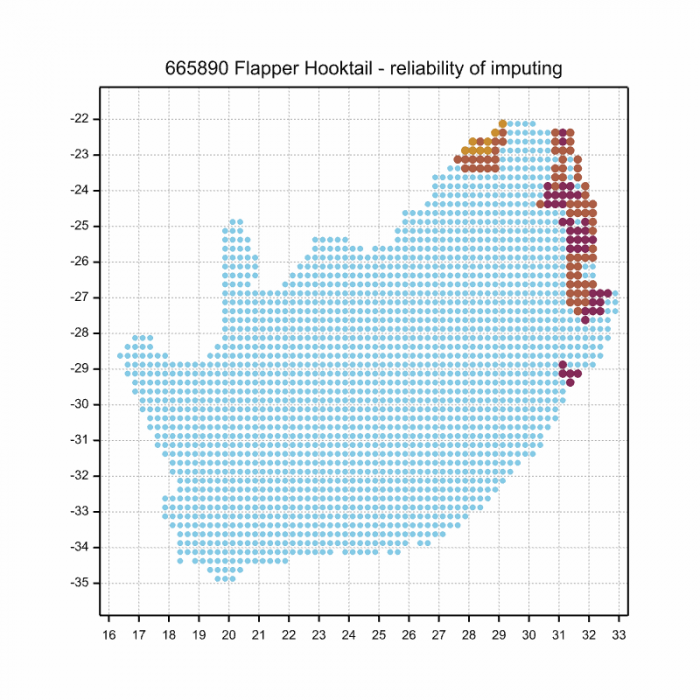
Ultimately, we will produce a series of maps for all the odonata species in the region. The current algorithm is a new algorithm. The objective is mainly to produce “smoothed” maps that could go into a field guide for odonata. This basic version of the algorithm (as mapped above) does not make use of “explanatory variables” (e.g. altitude, terrain roughness, presence of freshwater — we will be producing maps that take these variables into account soon). Currently, it only makes use of the OdonataMAP records for the species being mapped, as well as all the other records of all other species. The basic maps are “optimistic” and will generally show ranges to be larger than what they probably are.
These maps use the data in the OdonataMAP section of the Virtual Museum, and also the database assembled by the previous JRS funded project, which was led by Professor Michael Samways and Dr KD Dijkstra.
Phenology


Further Resources
The use of photographs by Desire Darling is acknowledged. All other photographs by Ryan Tippett.
Flapper Hooktail Paragomphus sabicus (Pinhey, 1950)
Other common names: Sabi Hooktail, Clubbed Hooktail (Alt. English); Flaphakiestert (Afrikaans)
Recommended citation format: Loftie-Eaton M; Navarro R; Tippett RM; Underhill L. 2025. Flapper Hooktail Paragomphus sabicus. Biodiversity and Development Institute. Available online at https://thebdi.org/2020/06/08/flapper-hooktail-paragomphus-sabicus/
References: Tarboton, M; Tarboton, W. (2019). A Guide to the Dragonflies & Damselflies of South Africa. Struik Nature.
Samways, MJ. (2008). Dragonflies and Damselflies of South Africa. Pensoft
Samways, MJ. (2016). Manual of Freshwater Assessment for South Africa: Dragonfly Biotic Index.Suricata 2. South African National Biodiversity Institute, Pretoria
Martens, A; Suhling, F. (2007). Dragonflies and Damselflies of Namibia. Gamsberg Macmillan.