Welcome to the September 2023 edition of the BDInsight. We have plenty of news to share and plenty of exciting events coming up. Read on to find out more…..
Biodiversity Observations
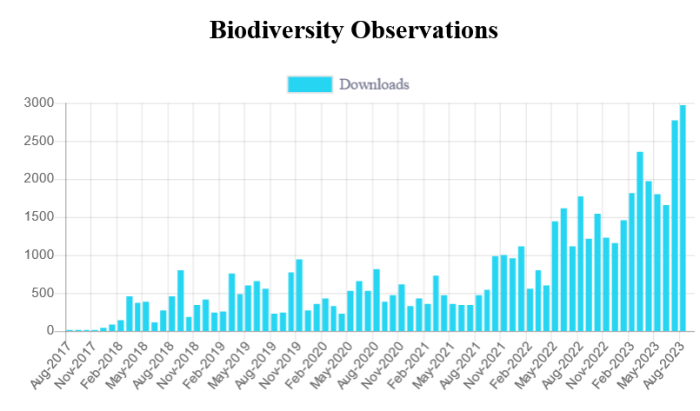
The graph shows that there were 2978 downloads of papers in Biodiversity Observations during August 2023. That is a new record! In 2023, we are working on Volume 13, and there have been 28 papers published this year so far, with a total of 232 pages.
Of the papers published in 2023, the one with the most downloads is A Guide to the Common Garden Birds of Cape Town, South Africa which had been downloaded 1172 times by 31 August. You can find the the paper here: https://journals.uct.ac.za/index.php/BO/article/view/1316. This link does not download the paper itself; it takes you to the page where you click on the PDF to get the paper. The image below is the Common Waxbill page in the garden birds guide.

12 out of the 28 papers in Biodiversity Observations so far in 2023 deal with observations on birds in South Africa. This means that the journal is getting increasing support from other taxa and from other countries (and continents). For example there is an excellent paper by Mike Fraser on the reptiles and amphibians of the Cape of Good Hope section of the Table Mountain National Park (here), two papers on ducks in Algeria (here and here), one on a bullfinch on an island in the Caribbean (here), one on a snail in Turkey (here), etc. Here is a list of the 28 papers in 2023 volume to date: https://journals.uct.ac.za/index.php/BO/index
A key measure of the success of a journal is when other authors cite papers in it. One of the most fascinating papers in the 2022 volume last year described the havoc caused by an invasive snail in India. The Giant African Snail Lissachatina fulica is classified as one of 100 worst invasive alien species and has been introduced to more than 50 countries. These are huge snails and they are indigenous to Africa (believe it or not this snail gets kept as a pet). The Biodiversity Observations paper has been cited in a paper about this snail into Italy. The link to the original paper is here; the paper from Italy that cites it is here (and the new paper is in an Open Access journal).

The first paper in the volume 13 of Biodiversity Observations provides a summary of the first 12 volumes! It is available here.
BDI Bird Species Identification Texts
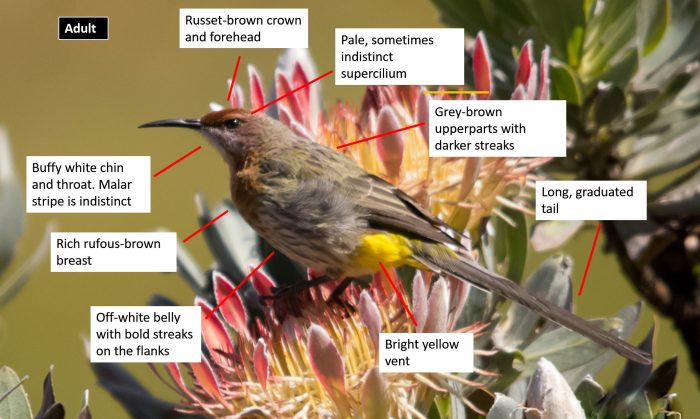
Species identification consists mostly of moving backwards and forwards between an illustration, and text describing the key identification features. We have simplified that, and put the text onto the illustration. The image here is for the Karoo Lark:

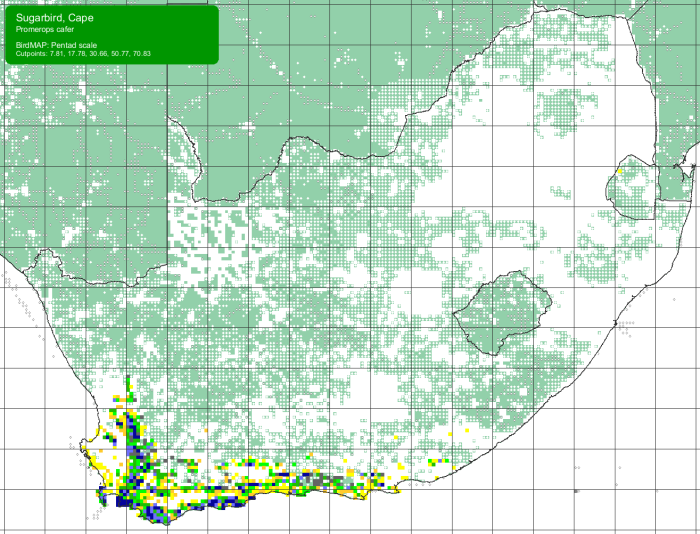
There are now 75 “BDI-style” species texts for birds on our website. Each of the texts starts with an annotated photograph like the one above. The next sections cover Habitat (with photos), Distribution (with bird atlas map), and Behaviour (with words and photos).
The website links for the 75 species which we have done to date can be found here: http://thebdi.org/bird-species/. Click on the name of any species on this page, and you get the BDI text for it. This is what the website looks like:
If you do a google search for any of these 75 species, the BDI text is usually on the first page of the search. Here is what to look out for in a google search for a species.

These species texts are available from the Virtual Museum. The Virtual Museum record for a species which has been done, has the word “Text” on the second line. Click on it, and you get the BDI species text. For example, this is a recent record of Cape Sugarbird:

https://vmus.adu.org.za/?vm=BirdPix-258842
We are not doing the species at random. Our first target group of 32 species consisted of garden birds of Cape Town, and they were used to produce “A Guide to the Common Garden Birds of Cape Town”. As described above, this is the most downloaded paper in Biodiversity Observations so far this year. The species texts currently being produced “BDI-style” are for other similar themes. Watch this space.
With each BDInsight we will list the species with new texts.
Bird Ringing
We have learnt a huge amount about birds through bird ringing. Watch Mark Brown’s outstanding contribution called “Why ring birds?” to Conservation Conversations
There is also a blog on this topic on the BDI website. It is called “The value of bird ringing to conservation and research.” It is recommended reading!
Among other initiatives, the BDI is focusing lots of attention on bird ringing and training bird ringers this past year. The bottom line is that there is still a vast amount to learn about our birds which can most readily be done through bird ringing. There are two quantities that are crucial for understanding species, and for designing management plans for them. One is survival and the other is movement. For both of these, we know very little for almost every South African species.
The way to find out these quantities is to focus ringing effort for a long time on a small number of sites. This is what we plan to do.
The blog on the BDI website contains links to past ringing events and courses, going back to 2019: http://thebdi.org/2023/07/14/bdi-bird-ringing-courses-and-events/ It is a fascinating account of how our thinking has grown and matured over the past five years (and how we have persevered with this vision in spite of all the hassles).
You can find out when the next ringing courses and events are taking place here: http://thebdi.org/upcoming-bdi-events/. You are warmly invited to participate.
Virtual Museum
Up-to-date distribution maps for species are critical for taking conservation decisions about species. Spring is springing, and the butterflies and moths, dragonflies and damselflies, will soon be out and about. Now is the time to dust off your cameras and get out into the field and start refreshing records in the Virtual Museum.
Records made long ago in a grid cell are slowly losing their value as evidence that the species is still present there, and need to be refreshed.
If your access to the Virtual Museum is not working (eg password issues) please contact Megan Loftie-Eaton for help (megan@thebdi.org).
RAVE 2023!
RAVE = RINGING, ATLASING AND VIRTUAL MUSEUMING EXPEDITION
What are the dates, places and costs?
- 27 November–03 December at Botuin, Vanrhynsdorp
- 4–10 December at Vondeling Wine Farm, Paardeberg, Wellington
- 11–15 December at Ouberg Private Nature Reserve, Montagu
- 16–18 December at Botuin, Vanrhynsdorp
We invite you to attend this citizen science event for as many days as are feasible for you. The cost for food and accommodation (sharing) is R750 per person per day. At Botuin, single accommodation is available at a facility close by. There is one single room at Vondeling. The cost of a single room is R900 per person per day. These dates are confirmed and the accommodation booked, but they can be extended, either by operating at more than one place at the same time, or by starting earlier
What will you gain and what will you give?
As a participant, the most important ingredient towards the success of the RAVE is enthusiasm!

This RAVE is the initial major event for setting up the BioObservatory. Our specific goals during the RAVE focus on ringing, atlasing and the Virtual Museum (VM). Through bird ringing, atlasing, and BirdPixing (for the Virtual Museum), we aim to:
- obtain annual survival estimates of key species via recaptures of ringed birds.
- build information on local movements of birds by systematically ringing at nearby sites.
- strengthen our understanding of moult.
- improve our knowledge of annual breeding success and the timing of breeding through a monitoring programme that estimates the percentage of juveniles in the populations at each site.
- develop guides that will help ringers and trainee ringers to age and sex birds.
These goals will not be attained through a single expedition, but with each event we are laying the foundations for a BioObservatory. RAVE goals contribute towards the wider goals of the BioObservatory:
- We aim to increase the numbers of ringed birds at the three sites (here is a summary of BDI ringing events, with many having been held at the three RAVE sites). We are aiming to increase the size of the existing database. Ultimately, the purpose is to improve the conservation outlook for as many of the species at these sites as possible. We aim to achieve this using the information and understanding gained from bird ringing.
- We want to build on the existing bird atlas data for the three sites, with a view to moving in the direction of systematic atlasing, as has been shown to be dramatically effective in monitoring bird population sizes in the Hessequa Region, centred on Stilbaai. For waterbirds, we will supplement the atlasing by undertaking counts of waterbirds at nearby wetlands.
- We plan to do as comprehensive surveys as we can for the other taxa represented in the Virtual Museum, but especially butterflies and moths, dragonflies and damselflies, and dung beetles. We will try to make substantive contributions to all sections of the Virtual Museum (digitally curated photographs of various taxa). We need to work out monitoring strategies for these taxa which are appropriate for the three sites.
- We would like to engage with local communities. We want to share our enthusiasm for biodiversity with the people who live in the areas close to our sites. We need to brainstorm how to achieve this. It could be through an Open Day, when anyone can join us in ringing or any other activity. Alternatively, we could invite the teachers at a local school or members of the farming community to join us.
Where will we stay?
At Botuin there are three cottages that each sleep two people. There is unlimited accommodation close by. We have all our meals at Botuin. Botuin lies on the southeastern edge of Vanrhynsdorp, 300 km north of Cape Town (three hours’ drive) along the N7 towards Namibia. It is on the edge of Namaqualand.

At Vondeling there are two comfortable cottages in the werf. One sleeps six people in two bedrooms for three people each. The other, which we also use as our main gathering place, sleeps five: two bedrooms each with a double bed, and one with a single bed. A couple of tents and caravans are feasible. Vondeling Wine Farm is 75 km northeast of Cape Town (one hour), on the eastern edge of the Paardeberg.

At Ouberg there is accommodation for 11 people. There is no alternative accommodation in easy striking distance, but a handful of tents and caravans are eminently feasible. Ouberg is 205 km east of Cape Town (2.5 hours), in the mountains on the edge of the Little Karoo. It is 20 km north of Montagu.

Who is in charge?
The expedition will be led by Les Underhill (overall coordination), Dieter Oschadleus (bird ringing), Salome Willemse (administration) and Jean Ramsay (finances). We are supported in multiple areas by other citizen scientists.
How do I apply to join the RAVE?
Send an email to Salome Willemse (ring@thebdi.org), and tell her the dates you would like to attend. We will do our best to accommodate you.
When will the next RAVE take place?
We don’t know. It all depends on the success of this one! But our biggest fear is that we won’t have the capacity to meet the demand.
We hope that you will join us, either now or in the future!