Cover image: Malachite Sunbird by Pamela Kleiman – Steytlerville, Eastern Cape – BirdPix No. 10960
Identification
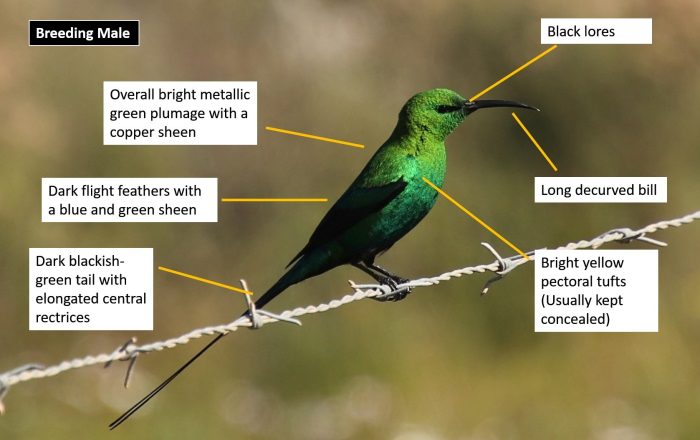
The Malachite Sunbird is notable for being the largest sunbird in southern Africa. The adult male in breeding plumage is highly distinctive and unmistakeable.
Near Darling, Western Cape
Photo by Marius Meiring
The overall plumage is bright metallic green with a copper sheen when seen in bright sunlight. The rump and upper tail coverts are slightly darker green. The tail is dark blackish-green with a blue gloss and the central tail feathers are elongated. The feathers in the folded wings are blackish with a green and blue metallic sheen. Displaying males have bright yellow pectoral tufts but these remain concealed at other times. The lores between the eyes and bill are black. The bill is very long and decurved. The bill and legs are black.

Steytlerville, Eastern Cape
Photo by Gregg Darling
Females are olive-grey above and paler grey-brown to yellow-brown below. The pale underparts contrast with the darker upperparts. Females have a pale yellowish supercilium and malar stripe. The tail is dark brown with white outer tail feathers and lacks the elongated central rectrices of the breeding males.

De Kelders, Western Cape
Photo by Sybrand Venter
Non-breeding males in eclipse plumage resemble females but have variable green-blotches on the underparts and wings. Juveniles closely resemble the females.

Mpofu Dam, Eastern Cape
Photo by Gregg Darling
Status and Distribution
The Malachite Sunbird is a locally common to common resident and partial migrant. Its distribution is discontinuous from central Ethiopia south through east Africa to South Africa. In South Africa the Malachite Sunbird ranges from central Limpopo Province south through Mpumalanga and western Swaziland to KwaZulu-Natal, and down to the Western Cape and up to Namaqualand and extreme southern Namibia. There is also an isolated population in the eastern highlands of Zimbabwe and adjacent Mozambique.
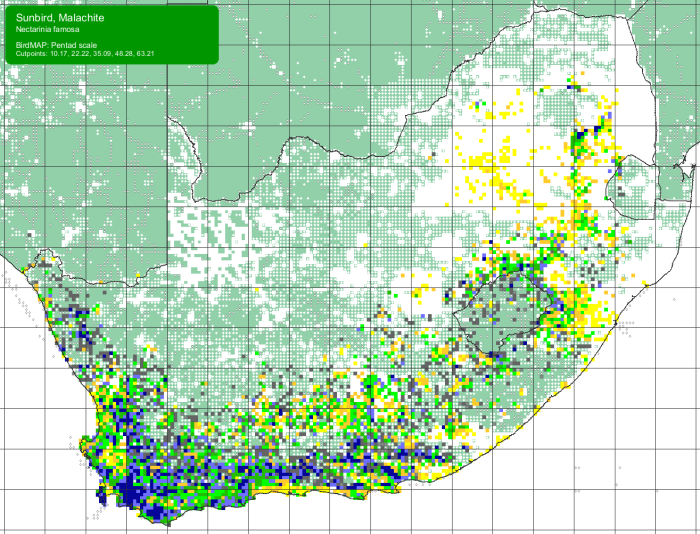
Details for map interpretation can be found here.
In South Africa the Malachite Sunbird appears to concentrate in the east of its range during the summer and in the west during winter. It generally moves out of the drier western areas (winter rainfall) in summer when it is hot and dry and flowering plants become scarce. This sunbird is an altitudinal migrant in the high altitude regions, particularly the Drakensberg, and moves to warmer, lower lying areas during the winter months.
There is no evidence of significant changes to its distribution or abundance. The Species has benefited from the planting of garden ornamentals which may have allowed the Malachite Sunbird to expand its range. In some areas, agriculture, forestry, urban development and alien plant infestations have impacted on its natural habitat. The Malachite Sunbird is not considered threatened.
Habitat
The Malachite Sunbird occupies a range of habitats from fynbos to mountain grasslands, forest edge and karoo scrub. It is often associated with scrubby hillsides and riverine bush, as well as parks, gardens and alien plantations. As a primarily nectarivorous species, its distribution and abundance are determined by the flowering cycles of food plants.

Golden Gate Highlands National Park, Free State
Photo by Ryan Tippett
The Malachite Sunbird is most common in montane grasslands and fynbos, followed by the Succulent Karoo and the southern parts of the Nama Karoo. It generally avoids the central and northern regions of the Nama Karoo which is more grass dominated and offers little food for sunbirds.
Behaviour
The Malachite Sunbird is an active and conspicuous species. It is usually encountered singly or in pairs when breeding. Gathers in large numbers at localised, rich food sources outside of the breeding season. Malachite Sunbirds may get displaced by Cape Sugarbird (Promerops cafer), Gurney’s Sugarbird (Promerops gurneyi) and the more aggressive Bronzy Sunbird (Nectarina kilimensis), but males regularly chase conspecifics and other, smaller sunbirds.

Somerset West, Western Cape
Photo by Corrie du Toit
The diet consists mostly of nectar and is supplemented with arthropods, especially spiders and small insects like flies, moths, caterpillars, bugs and beetles. Insect prey is mostly gleaned from vegetation but the Malachite Sunbird will also hawk flying insects from time to time. Some pollen is also eaten and they readily take sugar water mixes from garden nectar feeders.
The Malachite Sunbird feeds on the nectar from a diverse array of plant species. The flowers of various Protea, Aloe, Leonotis and Kniphofia species are particularly important, and this sunbird is a key pollinator of these plants.

Waaihoek Mountains, Western Cape
Photo by Fanie Rautenbach
Bathes in rain-soaked foliage, bird baths and in irrigation sprays. The flight is fast and direct.
An interesting adaptation of the Malachite Sunbird is its ability to enter torpor (daily hibernation) at night. This saves up to 60% of energy by reducing body temperature to as low as 26°C.

Malachite Sunbird (Nectarina famosa)
Near Durbanville, Western Cape
Photo by Gerald Wingate
The Malachite Sunbird breeds from May to November in the winter rainfall Western Cape, and during the summer months (mainly October to January) over the rest of its range. The nest is an oval of grass, leaves, twigs and roots, bound together with spider web. The nest has a side-top entrance and the inside is lined with fine grass, hair, feathers and plant fibres. The entrance usually has an overhanging ‘porch-style’ roof. The nest is often placed over a stream, gully or cliff face and is built entirely by the female.

Hermanus, Western Cape
Photo by Andries de Vries
One to three (usually two) eggs are laid per clutch. Incubation lasts for around 13 days and is performed solely by the female. The nestling period takes about 20 days, during which time the young are fed and cared for by both parents.
Malachite Sunbird nests are frequently parasitised by Klaas’s Cuckoo (Chrysococcyx klaas), and to a lesser extent by Red-Chested Cuckoo (Cuculus solitarius) and Diederik Cuckoo (Chrysococcyx caprius).
Further Resources
Species text adapted from the first Southern African Bird Atlas Project (SABAP1), 1997.
The use of photographs by Andries de Vries, Cobus Elstadt, Corrie du Toit, Fanie Rautenbach, Gerald Wingate, Gregg Darling, Marius Meiring, Pamela Kleiman and Sybrand Venter is acknowledged.
Virtual Museum (BirdPix > Search VM > By Scientific or Common Name).
Other common names: Jangroentjie (Afrikaans); iNcuncu, iNcwincwi, uHlazazana (Zulu); Tale-tale (South Sotho); Souimanga malachite (French); Emeraldhoningzuiger (Dutch); Malachitnektarvogel (German); Beija-flor-verde (Portuguese).
List of bird species in this format is available here.
Recommended citation format: Tippett RM 2024. Malachite Sunbird Nectarina famosa. Biodiversity and Development Institute. Available online at https://thebdi.org/malachite-sunbird-nectarina-famosa/

Uniondale district, Western Cape
Photo by Cobus Elstadt